Symphylans: Opciones de manejo de plagas del suelo
Por Jon Umble, Universidad Estatal de Oregón; Rex Dufour, NCAT; Glenn Fisher, Universidad Estatal de Oregón; James Fisher, USDA/ARS; Jim Leap, Universidad de California, Santa Cruz; Mark Van Horn, Universidad de California, Davis

Un simpílico de jardín es aproximadamente del tamaño de esta letra “l”.
Resumen
Los sínfilos de jardín son criaturas parecidas a ciempiés que habitan en el suelo y que se alimentan de las raíces de las plantas y pueden causar daños extensos en los cultivos. Causan problemas frecuentes y a menudo mal diagnosticados en suelos occidentales bien manejados con buena labranza. Esta plaga del suelo puede no ser familiar para los agricultores y consultores agrícolas. Esta publicación describe el ciclo de vida del sínfilo del jardín y el daño que pueden causar los sínfisianos. Incluye técnicas de monitoreo para determinar si los sínfilos están presentes en el suelo y opciones de manejo sostenible para prevenir daños económicos.
Contenido
Introducción
Daño
Identificación
Biología y Ecología
Muestreo
Gestión y Control
Referencias
Introducción

Suelos con alta materia orgánica, buena estructura, y
La reducción de la perturbación, como en estas camas de jardín excavadas a mano, es un hábitat ideal para los sínfilos de jardín.
Los sínfilos de jardín (Scutigerella immaculata Newport) son pequeños artrópodos de suelo blancos, parecidos a ciempiés, comunes en muchos sistemas de producción agrícola en Oregón, Washington y California (Berry y Robinson, 1974; Michelbacher, 1935).
Se alimentan de raíces y otras partes subterráneas de plantas, causando pérdidas significativas de cultivos en algunos casos. El control puede ser extremadamente difícil debido al movimiento vertical de los sínfilos en el suelo, la complejidad del muestreo y la falta de métodos de control simples y efectivos (Umble y Fisher, 2003a).
Con la reciente difusión de la agricultura orgánica y mejores técnicas de manejo del suelo, el daño a los cultivos asociado con los sínfilos se ha vuelto más común. Es irónico que estas plagas sean un problema en las granjas que practican un buen manejo del suelo: mantener el suelo con buena labranza, alta materia orgánica y baja compactación.
Daño
Diagnosticar un problema de sinfílico de jardín a veces es difícil, ya que el daño puede exhibirse en varias formas y los sínfilos de jardín no siempre son fáciles de encontrar, incluso cuando su daño es obvio. El daño económico puede resultar de la alimentación directa de cultivos de raíces y tubérculos y la reducción de rodales de cultivos directos sembrados o trasplantados (Umble y Fisher, 2003a).
Sin embargo, más comúnmente, la alimentación de raíces reduce la capacidad del cultivo para absorber agua y nutrientes, lo que conduce a un retraso general en el crecimiento. El daño de las raíces también puede hacer que las plantas sean más susceptibles a algunos patógenos de plantas transmitidos por el suelo. Diagnóstico correcto de
Los problemas de sinfílico de jardines y la identificación de tácticas de manejo apropiadas para un sistema de cultivo dado generalmente requerirán lo siguiente:
- Muestreo para determinar si los sínfilos de jardín están presentes en números dañinos
- Un conocimiento general de las tácticas de manejo y la ecología del sínfilo del jardín para seleccionar las tácticas específicas que serán más efectivas en un sistema de cultivo determinado.

Berenjena atrofiada por sínfilos de jardín.

Berenjena intacta de la misma edad en el mismo campo.

El daño por symphylan puede ser difícil de diagnosticar porque puede parecer que se debe a otras causas. Esta foto muestra dos filas de berenjenas, una que está severamente atrofiada y otra menos. Ni las filas a la izquierda ni los pimientos de la derecha parecen verse afectados. Es probable que el momento de la siembra y la labranza influyan en la cantidad de daño que causan los sínfilanos. Muchos factores pueden interactuar para promover o reducir el daño del sínfisilano: condiciones cuando se cultiva el suelo, tiempo después de la labranza, requisitos de calor del cultivo, manejo del riego, tamaño de la planta, etc.


Daño típico del sínfilo desconcertante: Algunas filas de pimientos (arriba, arriba) están severamente atrofiadas, mientras que las filas adyacentes tienen plantas sanas y atrofiadas. El daño de Symphylan puede confundirse con la siembra omitida o el contacto deficiente de la semilla con el suelo, como en estos campos de calabaza (arriba, abajo), maíz dulce y tomates (abajo)



Ciertas áreas de este campo de squash son destruidas por symphylans, mientras que otras secciones prosperan.

Daño típico de symphylan, que muestra plantas sanas junto con plantas atrofiadas y áreas vacías.
Identificación
Los sínfilos de jardín no son insectos, sino miembros de la clase Symphyla. Las especies de esta clase son artrópodos comunes del suelo en todo el mundo. Symphyla son criaturas pequeñas, blanquecinas parecidas a ciempiés que van desde menos de 1/8 de pulgada hasta aproximadamente 5/8 de pulgada (o 1/4 de pulgada para los simfilanes de jardín) (Edwards, 1990). Tienen de seis a 12 pares de patas (dependiendo de la edad), lo que los hace fáciles de distinguir de los insectos comunes del suelo (por ejemplo, colémbolos) y dipluranos que tienen solo tres pares de patas, todas en el tórax o segmento medio del cuerpo.
Aunque su color puede variar, dependiendo de lo que hayan comido, los sínfilos de jardín son generalmente más blancos y más pequeños que los verdaderos ciempiés, que también son artrópodos del suelo con muchos pares de patas (un par por segmento corporal) y movimientos rápidos. Los milpiés generalmente se mueven más lentamente, con dos pares de patas en cada segmento del cuerpo.
Algunas especies de Symphyla se alimentan principalmente de materia orgánica muerta o en descomposición, desempeñando un papel importante en el ciclo de nutrientes. Otras especies, como el attsymphylan de jardín, son plagas graves, alimentándose principalmente de plantas vivas.
Varias especies de Symphyla están presentes en el oeste de los Estados Unidos; sin embargo, el simpylán de jardín es la única especie de Symphyla que está documentada para causar daños a los cultivos en el oeste de EE.UU. Los symphylans de jardín son, con mucho, las especies de Symphyla más comunes que se encuentran en los sistemas agrícolas.
Si Symphyla se encuentra en un sistema agrícola de alta densidad, concentrado alrededor de las raíces de las plantas, es probable que sean sínfilos de jardín. No se sabe que la plaga vectorice ninguna enfermedad de las plantas, aunque no se ha realizado una investigación exhaustiva sobre la cuestión.
Jardín Symphylan Biología y Ecología
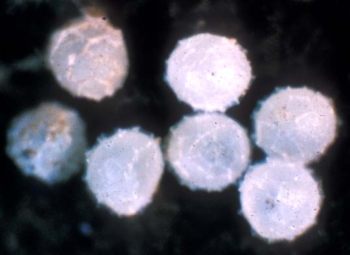
Huevos de sínfilo de jardín. Fotografía: Ralph Berry
Ciclo vital
En el oeste de los Estados Unidos, los huevos, los adultos y los sínfilos de jardín inmaduros se pueden encontrar juntos durante la mayor parte del año. La temperatura juega un papel clave en la regulación de la oviposición (puesta de huevos), y el mayor número de huevos generalmente se depositan en la primavera y el otoño (Berry, 1972).

Symphylans recién emergidos (primer estadio). Crédito de la foto: Ralph Berry
Los huevos son de color blanco nacarado y esféricos con crestas de forma hexagonal. Los huevos se incuban durante aproximadamente 25 a 40 días, cuando las temperaturas oscilan entre 50 ° y 70 ° F, pero la eclosión ocurre en aproximadamente 12 días a medida que las temperaturas alcanzan los 77 ° F (Berry, 1972).
Los primeros estadios emergen del huevo con seis pares de patas y seis segmentos antenales, sus cuerpos cubiertos de pelos finos. Los movimientos lentos y una parte posterior hinchada hacen que los primeros estadios parezcan superficialmente más un colémbolo que un sínfilo de jardín adulto. Estos primeros estadios, sin embargo, rara vez se encuentran en la zona de enraizamiento y en cuestión de días mudan a segundos estadios que se asemejan a pequeños sínfilos de jardín adultos (Michelbacher, 1938).

Sínfilo de jardín adulto maduro.
Cada una de las seis mudas posteriores da como resultado la adición de un par de patas y un número variable de segmentos corporales y antenas. El tiempo total desde el huevo hasta el adulto sexualmente maduro (séptimo estadio) es de aproximadamente cinco meses a 50 ° F, disminuyendo a aproximadamente tres meses a 70 ° F y menos de dos meses a 77 ° F. Por lo tanto, puede ser posible tener dos generaciones completas al año (Berry, 1972). Curiosamente, a diferencia de los insectos adultos, que no mudan, los sínfilos de jardín adultos pueden mudar más de 40 veces (Michelbacher, 1938).
Ocurrencia
La comprensión de la ocurrencia y el movimiento del sínfilo del jardín está lejos de
íntegro. No obstante, se pueden hacer algunas generalizaciones tanto sobre los suelos como sobre los suelos en
qué sínfilos de jardín ocurren más comúnmente y sobre su movimiento en
Suelos. Las poblaciones de sínfilos de jardín están altamente agregadas dentro de los campos y
a mayor escala.

Los campos a menudo muestran daños por sínfila en los mismos lugares durante muchas estaciones, como en estas dos granjas.

En Oregón, Washington y California, los sínfilos de jardín son más comunes en las regiones occidentales de los estados. Dentro de estas regiones, los sínfilos de jardín tienden a ocurrir en suelos irrigados más pesados, y dentro de estos suelos más pesados, los sínfilos de jardín tienden a ocurrir en “puntos calientes” de unos pocos pies cuadrados a varios acres. Incluso dentro de palas de tierra, los sínfilos de jardín a menudo ocurren en agregaciones muy distintas.
Los sínfilos de jardín son incapaces de excavar a través del suelo. Utilizan poros, grietas estacionales y madrigueras hechas por otros animales del suelo, como las lombrices de tierra, para viajar a través del perfil del suelo (Edwards, 1961).

Los poros, grietas y agujeros en el suelo permiten síndicos
para moverse a través de un campo con relativa facilidad.
En general, las prácticas que mejoran la estructura del suelo (por ejemplo, adición de materia orgánica, labranza reducida, camas elevadas) mejoran la capacidad de los sínfilos de jardín para moverse a través del suelo, lo que lleva a un aumento de las poblaciones y / o un mayor daño a través de un mejor acceso a las raíces. Como resultado, las altas poblaciones de sínfilos de jardín se encuentran más comúnmente en suelos de textura fina, más pesados con estructura moderada o mejor y muchos macroporos, en lugar de en suelos arenosos (Edwards, 1958; Edwards, 1961). Cuando los sínfilos de jardín se encuentran en suelos más arenosos, estos suelos han sido comúnmente modificados con materia orgánica.
En el noroeste del Pacífico y el norte de California, los sínfilos de jardín se encuentran comúnmente en suelos aluviales, y es probable que se propaguen en cierta medida por inundaciones. La acidez relativa del suelo no parece estar estrechamente correlacionada con la ocurrencia de sínfilos de jardín, ya que los sínfilos se encuentran en suelos muy ácidos (por ejemplo, donde crecen los arándanos) a suelos bastante alcalinos (por ejemplo, pH 8+).

“Hotspots” del espectáculo de infestación de sinfílicos de jardín
claramente en esta vista aérea de un campo de brócoli.



Arriba: Tres vistas de un campo de brócoli que muestran daños por sínfisilano.
Los puntos calientes dentro de los campos infestados a menudo permanecen consistentes de año en año con pocos cambios en las poblaciones y solo una propagación lateral menor, posiblemente debido a las características físicas del suelo. Sin embargo, se producen cambios en los puntos críticos (Umble y Fisher, 2003c).
Movimiento de sínfilos en el suelo y factores que influyen en los niveles de población
Si el ambiente del suelo es favorable, los sínfilos del jardín pueden migrar desde la superficie del suelo a una profundidad de más de 3 pies. El perfil del suelo, incluidos los horizontes compactados o arenosos y las capas freáticas altas que pueden impedir el movimiento, determina la profundidad a la que pueden migrar los sínfilos de jardín. El momento de las migraciones verticales se debe principalmente a la interacción entre la humedad, la temperatura y los ciclos de alimentación regulados internamente (Edwards, 1959b). Una comprensión general de estas interacciones es importante tanto para el momento y la interpretación de los esfuerzos de muestreo como para seleccionar tácticas de gestión.
Los sínfilos de jardín tienden a agregarse en las seis pulgadas superiores del suelo cuando el suelo está húmedo y cálido, y se mueven a estratos de suelo más profundos cuando el suelo se vuelve muy seco o frío. En Oregón, Washington y California, los sínfilos de jardín se encuentran generalmente en el suelo superficial de marzo a noviembre, con las poblaciones de superficie más altas observadas en mayo y junio. Los sínfilos de jardín se pueden encontrar en la superficie del suelo cuando las condiciones son bastante cálidas (por ejemplo, cuando las temperaturas del aire exceden los 95 ° F), si hay suficiente humedad presente y las raíces son poco profundas o están ausentes. En los valles interiores más cálidos, los sínfilos pueden ser más activos en la primavera / principios del verano y el otoño, con la actividad superficial disminuyendo en julio, agosto y septiembre.
Los sínfilos de jardín migran al suelo superficial (la zona de la raíz) para alimentarse, luego regresan a los estratos más profundos para mudar, como lo demuestra la gran cantidad de pieles mudadas que se observan en estos estratos. Cuando los sínfilos del jardín se alimentan vorazmente después de la muda, pueden ingresar a la zona del suelo superficial incluso en condiciones generalmente desfavorables (por ejemplo, cálidas y secas). Dado que las migraciones no están sincronizadas, las porciones de la población generalmente están presentes en toda la porción habitable del perfil del suelo (Edwards, 1959b). La presencia de sínfilos de jardín en la superficie del suelo también puede estar influenciada por otras variables que impiden el movimiento, como la labranza y la compactación de los neumáticos de tractor.
Muestreo
El muestreo de sínfilos de jardín es extremadamente importante para identificar daños, para tomar decisiones de manejo informadas y para evaluar los efectos de esas decisiones. El muestreo, sin embargo, es a menudo difícil. Se utilizan tres métodos principales de muestreo: cebo, muestreo de suelo y muestreo indirecto. Cada método tiene ventajas e inconvenientes, y la selección de un método de muestreo variará, dependiendo de los objetivos del muestreo (por ejemplo, detección vs. estimación precisa de la densidad de población), la época del año y las condiciones del sitio.
Parte de la dificultad en el muestreo se debe a la distribución irregular de las poblaciones. Es importante tener en cuenta que un recuento de muestras individuales proporciona información solo sobre la región cerca de donde se tomó esa muestra. Los recuentos a menudo variarán de 0 a más de 50 sinfilanes de jardín por muestra. Para obtener información sobre los patrones espaciales de la población, es mejor tomar unidades de muestra en un patrón de cuadrícula. Clasificar y comparar las muestras por factores del sitio, como el tipo de suelo, el drenaje y el historial de cultivos, puede proporcionar información valiosa sobre la distribución de las poblaciones dentro de un sitio.
Directrices generales para la toma de muestras
Para detectar o identificar un problema de symphylan con un cultivo en crecimiento:
- Desentierra las plantas y malezas atrofiadas por la mañana; Examine sus raíces en busca de sínfilanos.
- Si es dentro de las tres semanas posteriores a la siembra, coloque cebos para sínfilos de jardín en áreas sospechosas de problemas.
Para estimar la densidad de población y/o tomar decisiones antes de plantar un cultivo:
- Tome muestras de suelo si el suelo está fresco o muy seco, si el campo está muy lleno de maleza o si está creciendo un cultivo de cobertura.
- Use muestras de cebo si el suelo es cálido y húmedo con vegetación escasa o si el suelo está desnudo.
En la mayoría de los casos, el muestreo mide solo la densidad de los sinfilanos en la superficie del suelo; Por lo tanto, el muestreo solo debe realizarse cuando los sínfilos de jardín están en el suelo superficial. Las mejores condiciones de muestreo son generalmente suelo cálido y húmedo. El muestreo dentro de las tres semanas posteriores a la labranza principal, como el disco, el arado o la pala, a menudo es inexacto, ya que los sínfilos del jardín pueden no haber tenido tiempo suficiente para restablecerse en el suelo superficial. Si el muestreo se realiza poco después de la labranza, se deben utilizar métodos de muestreo del suelo. El muestreo debe realizarse a una profundidad que incluya varias pulgadas de suelo no perturbado por la labranza.
Muestreo de suelos
El muestreo de suelo es el método estándar/histórico para estimar cuántos sinfilanes de jardín están presentes en un campo (es decir, número aproximado de sinfilanes de jardín por unidad de suelo, o densidad de población estimada) (Berry y Robinson, 1974). Los tamaños de las unidades de muestra varían. Las unidades de muestreo de suelo más comunes han sido las siguientes:
- Un cubo de 1 pie
- Un cuadrado de 6 pulgadas, 1 pie de profundidad
- Una “pala”
- Núcleos de 3 a 4 pulgadas de diámetro y 4 a 12 pulgadas de profundidad

El muestreo del suelo en el maíz se lleva a cabo colocando tierra en una lona negra y luego buscando cuidadosamente los sínfilos del jardín.
Cuando se toman muestras de suelo, el suelo de cada unidad de muestra generalmente se coloca sobre un trozo de plástico oscuro o tela, donde los agregados se rompen y se cuentan los sínfilos del jardín (Berry y Robinson, 1974).
El muestreo debe realizarse en toda la región habitable del perfil del suelo (es decir, posiblemente a una profundidad de más de tres pies) para obtener estimaciones precisas de la densidad de población, pero esto rara vez se hace, debido al extenso tiempo y recursos requeridos. Por lo tanto, el muestreo generalmente se realiza cuando se cree que los sínfilos del jardín están en las 6 a 12 pulgadas superiores de la zona de la raíz. El muestreo superficial (por ejemplo, a una profundidad de 4 pulgadas) ahorra tiempo y permite muestrear áreas más grandes, pero el muestreo más profundo (por ejemplo, a una profundidad de 12 pulgadas) generalmente es más confiable. No se recomienda el muestreo en condiciones muy secas.
Muestreo de cebo
 En los últimos años, se han desarrollado métodos de muestreo de cebo. Las muestras de cebo son generalmente mucho más rápidas de tomar que las muestras de suelo, pero también son más variables y más sensibles a factores como la humedad del suelo, la temperatura y la presencia de vegetación (Umble y Fisher, 2003b). Para cebar la muestra, coloque la mitad de una papa o remolacha en la superficie del suelo y protéjala con una cubierta protectora (por ejemplo, una maceta blanca o una tapa de PVC de 4 pulgadas).
En los últimos años, se han desarrollado métodos de muestreo de cebo. Las muestras de cebo son generalmente mucho más rápidas de tomar que las muestras de suelo, pero también son más variables y más sensibles a factores como la humedad del suelo, la temperatura y la presencia de vegetación (Umble y Fisher, 2003b). Para cebar la muestra, coloque la mitad de una papa o remolacha en la superficie del suelo y protéjala con una cubierta protectora (por ejemplo, una maceta blanca o una tapa de PVC de 4 pulgadas).
 De uno a tres días después de la colocación, levante el cebo y cuente primero los sínfilos de jardín en el suelo y segundo los sínfilos de jardín en el cebo. Durante condiciones cálidas y / o secas, los cebos generalmente se revisan uno o dos días después de la colocación, ya que los recuentos disminuyen si los cebos se dejan fuera durante varios días. En condiciones más frías, los cebos se dejan comúnmente fuera durante tres a cinco días.
De uno a tres días después de la colocación, levante el cebo y cuente primero los sínfilos de jardín en el suelo y segundo los sínfilos de jardín en el cebo. Durante condiciones cálidas y / o secas, los cebos generalmente se revisan uno o dos días después de la colocación, ya que los recuentos disminuyen si los cebos se dejan fuera durante varios días. En condiciones más frías, los cebos se dejan comúnmente fuera durante tres a cinco días.

Fotos arriba: Usando una papa cortada como cebo para verificar la presencia de sínfilos de jardín.
El muestreo de cebo funciona muy bien para algunas aplicaciones, aunque no se puede usar en todas las condiciones. El cebo funciona mejor al menos dos o tres semanas después de la labranza, cuando el suelo se ha estabilizado, pero antes de que las plantas estén bien establecidas. Si viajar al lugar de muestreo requiere recursos significativos, se pueden preferir los métodos de muestreo de suelo, ya que requieren solo un viaje al sitio.
Métodos de muestreo indirecto
El crecimiento de las plantas a veces puede ser una medida indirecta útil de las poblaciones de sinfílicos de jardín y a menudo es un buen punto de partida para evaluar sus patrones espaciales (Umble y Fisher, 2003a). Por ejemplo, las plantas sanas a veces indican poblaciones de sinfílicos de jardín bajas y, a la inversa, las plantas no saludables a veces indican poblaciones de sínfilos de jardín altas.
¿Cuántas unidades de muestra de suelo o murciélagos usar?
Al muestrear sínfilos de jardín, se requiere un número crítico de unidades de muestra (es decir, “trozos” de tierra o cebos) para tener un nivel razonable de confianza sobre la densidad de población estimada (por ejemplo, sínfilos de jardín por pie cuadrado) (Umble y Fisher, 2003b). La confianza en esta estimación aumenta a medida que se toman más muestras. El muestreo implica establecer un equilibrio entre querer estar bastante seguro sobre el número de sinfilanes de jardín presentes (tomar un gran número de muestras) y no invertir demasiado tiempo y energía en el esfuerzo de muestreo (tomar un pequeño número de muestras).
Utilice las siguientes directrices generales para determinar el número adecuado de unidades de muestra.
- Simplemente detectar la presencia de sínfilos de jardín solo puede requerir excavar de cinco a 10 plantas dañadas, o usar un número bajo de cebos (por ejemplo, cinco)
- El muestreo para densidades de población bajas (a principios de la primavera o en cultivos altamente susceptibles) requiere más unidades de muestra (por ejemplo, 100+) que el muestreo para altas densidades de población (por ejemplo, 30)
- A medida que aumenta la variabilidad de un método de muestreo, también lo hace el número de unidades de muestra requeridas. Dado que el método de cebo es más variable que el método de muestreo del suelo, se requieren de dos a tres veces más cebos que muestras de suelo.
- Para estimar las densidades de población “económicamente dañinas” en cultivos moderadamente susceptibles, comúnmente se establecen al menos 35 unidades de muestra de suelo o al menos 50 muestras de cebo. Dependiendo del tamaño del campo y la época del año, se pueden usar considerablemente más unidades de muestra.
Medidas indirectas como esta pueden proporcionar información valiosa sobre el alcance y el patrón de la infestación, pero no deben utilizarse en lugar del muestreo directo. Esto se debe a que muchos factores podrían conducir a un crecimiento saludable de las plantas, incluso dentro del suelo infestado (por ejemplo, la fecha de siembra, la intensidad de la labranza, el uso de productos químicos y las especies de cultivo).


Las plántulas de espinaca muestran susceptibilidad al daño de los sínfisilos. El suelo en la maceta en la foto superior contiene 45 sinfilanos. El suelo en la maceta en la foto inferior no tiene sínfilas.
Umbrales de acción: interpretación de los resultados del muestreo
Las decisiones de manejo, como las relacionadas con las aplicaciones de pesticidas y la intensidad de la labranza, a veces se basan en estimaciones de densidad previas a la siembra de la población de sinfílicos de jardín. Debido en parte a los muchos cultivos en los que los sínfilos de jardín son plagas, los umbrales para cultivos individuales no están bien desarrollados. La relación entre las densidades de población de sínfilos de jardín (estimadas por métodos de muestreo) y medidas como el recuento de rodales y el rendimiento están influenciadas por factores como el tipo de cultivo, la intensidad de la labranza y la etapa del cultivo (Umble y Fisher, 2003a).
En el laboratorio, se ha demostrado que niveles tan bajos como cinco a 15 sínfilos de jardín por maceta reducen el crecimiento de plántulas de cultivos como frijoles, espinacas y maíz dulce (Umble y Fisher, 2003a; Eltoum y Berry, 1985). Se ha demostrado que la mayor densidad de 45 sínfilos de jardín por maceta reduce el crecimiento de tomates y plántulas de espinacas en más del 90%.
En el campo, a menudo se produce un daño notable si los sínfilos de jardín exceden un promedio de 5 a 10 por pala en cultivos de moderada a alta susceptibilidad como el brócoli, la calabaza, la espinaca y el repollo (Berry y Robinson, 1974; Umble y Fisher, 2003a).
En los sistemas de cultivo convencionales, dos o tres sínfilos de jardín por pie cuadrado se usan comúnmente como umbral de tratamiento.
Debido a la considerable variabilidad de las densidades de sínfilos dentro de un campo, los recuentos de unidades de muestra pueden variar de 0 a más de 100. Estos resultados son útiles para localizar puntos calientes de campo. En cultivos más tolerantes, como papas, frijoles y granos pequeños, la alimentación con sinfílico de jardín puede no provocar daños significativos, incluso a densidades de población considerablemente más altas (Umble y Fisher, 2003a).
Gestión y Control
Muchos factores influyen en los niveles de población de sínfilos de jardín (Howitt y Bullock, 1955; Umble y Fisher, 2003c; Getzin y Shanks, 1964; Shanks, 1966). Sin embargo, debido a que es difícil muestrear con precisión las poblaciones, la información sobre los verdaderos efectos de muchos factores en los sínfisicanos de jardín es, en el mejor de los casos, escasa. Para fines de manejo, es importante hacer una distinción entre las tácticas que disminuyen las poblaciones y las tácticas que reducen el daño a los cultivos, pero que no necesariamente disminuyen las poblaciones. En la mayoría de los casos, la gestión eficaz del sínfilo del jardín implica establecer un equilibrio entre estas dos estrategias. Es importante tener en cuenta que en la mayoría de los casos, después de que se nota el daño, poco se puede hacer sin replantar. Por lo tanto, el muestreo es importante para determinar el curso de acción adecuado.
Se desconoce si las poblaciones de symphylan pueden desarrollarse a partir de suelo transportado o compost. Estas son ciertamente posibles fuentes de infestación, y se recomienda tomar muestras de suelo y compost (dentro o fuera del sitio) para sínfilos antes de aplicar estas enmiendas a un campo. En general, se cree que las poblaciones de symphylan son de cosecha propia y se desarrollan con el tiempo debido a las prácticas favorables de manejo del suelo.
Tácticas para disminuir la población
La reducción de las poblaciones ha sido el foco de muchos estudios (Howitt y Bullock, 1955; Umble y Fisher, 2003c; Getzin y Shanks, 1964; Shanks, 1966; Howitt, 1959; Peachey y otros, 2002). Aunque no se han identificado “balas de plata”, algunas tácticas están disponibles. Probablemente ningún método erradicará los sínfilos de jardín de un sitio, y el efecto de la mayoría de las tácticas no durará más de uno a tres años.
Labranza
La labranza es probablemente la táctica de control más antigua, y sigue siendo una de las más efectivas (Martin, 1948; Peachey y otros, 2002). La labranza puede aplastar físicamente los sínfilos del jardín, reduciendo así las poblaciones. La labranza también puede dañar a las poblaciones de depredadores sinfilán de jardín clave, como los ciempiés y los ácaros depredadores. Sin embargo, en los sistemas de cultivo anual, los beneficios del aumento de las poblaciones de depredadores de la labranza reducida no han demostrado ser tan efectivos como la labranza en la disminución de las poblaciones de sínfilos de jardín. Al considerar el aumento de la labranza, los agricultores deben equilibrar los beneficios y los costos, como la materia orgánica oxidada del suelo, el suelo compactado, el aumento de los gastos de tiempo, combustible y equipo desgastado.
En general, para el control más efectivo, hasta cuando los sínfilos del jardín están en el suelo superficial y cuando la humedad del suelo permite la preparación de un lecho de semillas finas. Dado que solo una parte de la población de sínfilos se encuentra en la superficie del suelo, la labranza nunca proporciona un control completo. Sin embargo, las poblaciones de superficie son generalmente significativamente más bajas durante al menos dos o tres semanas después de la labranza.
Los sínfilos de jardín son criaturas parecidas a ciempiés que habitan en el suelo y se alimentan de raíces de plantas y pueden causar daños extensos, a menudo mal diagnosticados en los cultivos. Describe el ciclo de vida, las opciones de gestión sostenible.

Brócoli dañado (arriba) y no dañado (abajo) plantado al mismo tiempo en el mismo campo.


Pepinos dañados (arriba) y no dañados (abajo) plantados al mismo tiempo en el mismo campo.


Girasoles dañados (arriba) y no dañados (abajo) plantados al mismo tiempo en el mismo campo.


Pimientos dañados (arriba) y no dañados (abajo) plantados al mismo tiempo en el mismo campo.

Pesticidas
Cientos de compuestos se han utilizado contra los sínfilos de jardín en los últimos 100 años, con diferentes eficacias (Howitt y Bullock, 1955). Los fumigantes y los plaguicidas organofosforados han sido los más efectivos, pero muchos de estos
ya no están registrados. Los pesticidas pueden tener el efecto de matar los sínfilos del jardín y repelerlos de la superficie del suelo. No se ha demostrado que los pesticidas menos tóxicos (por ejemplo, piretroides y otros pesticidas naturales) proporcionen un control aceptable. Los pesticidas generalmente proporcionan la mayor cantidad de control cuando se transmiten e incorporan, aunque las aplicaciones con bandas e inyectadas pueden proporcionar un nivel aceptable de control.
Rotación de cultivos
Aunque los sínfilos de jardín se alimentan de una amplia gama de plantas, también pueden persistir en el suelo desnudo al alimentarse de otros organismos del suelo. Las plantas varían mucho en su susceptibilidad a los sínfilos de jardín. La rotación de cultivos puede explicar parcialmente los cambios aparentemente repentinos en los niveles de población de sinfílicos de jardín. Las poblaciones disminuyen significativamente en los cultivos de papa, incluso permitiendo el cultivo posterior de cultivos susceptibles en rotación (Umble y Fisher, 2003c). Aunque no hay otros cultivos han demostrado ser casi tan eficaces para reducir las poblaciones de sínfilos como las patatas, las poblaciones de sínfilos son más bajas después de un cultivo de cobertura de invierno de avena de primavera (‘Monida’) que después de un cultivo de cobertura de invierno de mostaza (‘Martiginia’), cebada (‘Micah’) o centeno (‘Wheeler’) (Peachey et al., 2002). Los cultivos de mostaza y espinacas son muy buenos huéspedes y pueden conducir a un aumento de las poblaciones en algunos casos. Todos estos factores deben considerarse al desarrollar un plan de manejo de malezas.
Otras enmiendas del suelo
Los efectos reportados de las enmiendas comunes del suelo, como el estiércol, la cal, los fertilizantes y el compost, varían mucho y a menudo son contradictorios. En general, se acepta que la cal y los fertilizantes tienen poco efecto en las poblaciones, mientras que generalmente se cree que las aplicaciones de estiércol aumentan las poblaciones (Shanks, 1966). El efecto del compost y las enmiendas orgánicas en las poblaciones de sinfílicos de jardín ha sido variable, pero en este momento no se ha demostrado que ninguno reduzca de manera consistente y significativa las poblaciones de simpulanos de jardín.
Tácticas para reducir el daño a los cultivos
La mayoría de las plantas pueden tolerar cierto nivel de alimentación de sinfílico de jardín durante toda o parte de la temporada de crecimiento, y dos tácticas generales pueden ayudar a cultivar cultivos saludables en suelos infestados de sínfilos. Estas tácticas son aquellas destinadas a reducir el daño a los cultivos cuando las poblaciones de sinfílicos de jardín son altas, y aquellas destinadas a reducir el número de sínfilos de jardín en las raíces de los cultivos durante el establecimiento, cuando las plantas son a menudo más susceptibles.
Tácticas para reducir el daño a los cultivos cuando las poblaciones de sinfílicos de jardines son altas

Papas plantadas bajo brócoli en UC Davis Student Farm.

Especie/variedad de cultivos
La susceptibilidad a la alimentación de sinfílicos de jardín puede variar dramáticamente entre diferentes especies y variedades de plantas. En la mayoría de los casos, la tolerancia a la alimentación parece deberse a un aumento del vigor y/o la producción de raíces (por ejemplo, brócoli, maíz) (Umble y Fisher, 2003a; Simigrai y Berry, 1974). En algunos casos, los sínfilos de jardín pueden simplemente comer menos de ciertos cultivos / variedades, aunque esto no se ha demostrado experimentalmente. En general, los cultivos de semillas más pequeñas tienden a ser más susceptibles que los cultivos de semillas más grandes (Umble y Fisher, 2003a). Los cultivos comúnmente dañados incluyen brócoli y otros cultivos de col, espinacas, remolachas, cebollas y calabazas.
Papas plantadas entre bloques de maíz dulce en prueba intercultivos en UC Santa Cruz Farm and Garden. Los frijoles y las papas rara vez se dañan, incluso bajo poblaciones altas. Los cultivos perennes, como las fresas, las frambuesas, el lúpulo y los árboles de raíz desnuda (producción de viveros), también pueden dañarse, particularmente durante el establecimiento. Dentro de una especie de cultivo, como el brócoli, algunas variedades son más tolerantes a los sínfilos de jardín que otras (Simigrai y Berry, 1974).

Los cultivos perennes también pueden ser dañados por los sínfilos que están presentes. Arriba: arándanos. Abajo: álamos híbridos.

Etapa de cultivo
Dentro de un cultivo, la susceptibilidad a menudo está relacionada con la etapa de desarrollo del cultivo. Por ejemplo, dentro de una variedad de tomate, los tomates de siembra directa son más susceptibles que los trasplantes de 4 semanas de edad, que son más susceptibles que los trasplantes de 12 semanas de edad. Usar trasplantes o aumentar el tamaño del trasplante para reducir. El daño no es efectivo para todos los cultivos. Los trasplantes de brócoli y berenjena, por ejemplo, a menudo no se establecen bajo poblaciones de sínfilos de jardín alto.
Densidad de plantas
Los sínfilos de jardín no cruzan la superficie del suelo por distancias significativas, como lo hacen los escarabajos de tierra. Sin embargo, son bastante activos y sorprendentemente móviles para su tamaño, moviéndose vertical y horizontalmente a través del perfil del suelo. Esto es sorprendentemente evidente cuando, por ejemplo, las plántulas trasplantadas en un semillero rancio con aparentemente pocos sínfilos de jardín tienen sínfilos de jardín arrastrándose por todas sus raíces menos de un día después de la siembra.
El número de sínfilos de jardín que se alimentan de cada planta en una región local (por ejemplo, lecho elevado) es parcialmente un factor del número de plantas presentes en ese lecho. En algunos casos, el aumento de la densidad de plantas, que por supuesto debe equilibrarse con las consideraciones de competencia de las plantas, produce una mejor producción. Las modificaciones de esta estrategia incluyen plantar un cultivo temprano de “distracción” o “dilución” en una cama o adyacente a una hilera de cultivo.
Un buen cultivo de dilución es un cultivo de bajo costo, vigoroso y fácil de establecer (por ejemplo, sudangrass en condiciones adecuadas) que aumenta las raíces en el suelo y efectivamente “diluye” los sínfilos del jardín lo suficiente como para establecer el cultivo objetivo. El cultivo de dilución se elimina a medida que se establece el cultivo objetivo.
Tácticas para reducir el acceso de los sínfilos de jardín a las raíces de los cultivos
Dado que los sínfilos de jardín no pueden excavar a través del suelo, sino que dependen de los poros y canales del suelo hechos por las raíces y otros organismos del suelo, su acceso a las raíces está fuertemente correlacionado con la estructura del suelo, la densidad aparente (“esponjosidad”) del suelo y la conectividad de los poros. En general, las siguientes tácticas se centran en reducir temporalmente el número de sínfilos de jardín en el suelo superficial antes de plantar, permitiendo así que los cultivos se establezcan mientras que los números de sínfilos de jardín son bajos.
Labranza
Además de matar directamente los sínfilos del jardín, la labranza rompe los agregados del suelo, modificando los poros del suelo y la conectividad de los poros. Los efectos de la labranza varían con los tipos de implementos utilizados. En general, cuanto más perjudicial sea la labranza, mayor será el efecto que tendrá en los sínfilos de jardín. El arado o el discar, seguido de una preparación minuciosa de un lecho de semillas finas utilizando un rototiller o roterra, a menudo reduce las poblaciones de sínfilos de jardín que se alimentan en la superficie durante dos o tres semanas. Durante este período de tiempo, los poros se forman a medida que se produce cierta agregación, y las lombrices de tierra y las raíces de las plantas forman nuevos canales a través del suelo. La alteración menos intensa del suelo, como la excavación manual o el cultivo poco profundo con una grada o un timón, puede tener un efecto significativamente menos perturbador en los sínfisios de jardín.

Plantas que crecen en el suelo compactado de las huellas de neumáticos en un campo desnudo e infestado de sínfilos.
Compactación/Camas elevadas
La protección de las raíces de las plantas de los sínfilos de jardín a veces es evidente en zonas donde los neumáticos de tractor han compactado el suelo, o en áreas donde un rototiller o disco ha formado una capa compactada o “bandeja de arado”.
Aunque la compactación puede tener algunos efectos negativos, en algunos suelos es posible compactar el suelo de manera beneficiosa utilizando, por ejemplo, un rodillo de paisajismo, reduciendo así el movimiento del sínfilo del jardín lo suficiente como para permitir que las plantas se establezcan. Las condiciones opuestas a menudo ocurren en camas elevadas que están altamente modificadas con materia orgánica, donde el suelo es muy bajo en densidad aparente y los sínfilos de jardín pueden moverse libremente a través de las camas.
Referencias
Berry, R. E. 1972. Sínfilo de jardín: Reproducción y desarrollo en el laboratorio. (Scutigerella immaculata). Revista de Entomología Económica. Vol. 65. págs. 1628-1632.
Berry, R. E. 1973. Biología del ácaro depredador, Pergamasus quisquiliarum, en el sínfilo de jardín, Scutigerella immaculata, en el laboratorio. Ann. Entomol. Soc. AM. 66: 1354-1356.
Berry, R. E., y R. R. Robinson. 1974. Biología y control del sínfilo de jardín. Universidad Estatal de Oregón. Servicio de extensión. Extensión Circular 845.
Edwards, C. A. 1957. Técnicas simples para criar Collembola, Symphyla y otros pequeños artrópodos que habitan en el suelo, pp. 412-416. En D. K. M. Kevan [ed .], Zoología del suelo. Butterworths Publications Ltd., Londres.
Edwards, C. A. 1958. La ecología de Symphyla: parte I. poblaciones. Entomologia Experimentalis et Applicata 1: 308-319.
Edwards, C. A. 1959a. Claves de los géneros de Symphyla. Revista de la Sociedad linneana XLIV: 164-169.
Edwards, C. A. 1959b. La ecología de Symphyla: parte II. Migraciones estacionales del suelo. Entomologia Experimentalis et Applicata 2: 257-267.
Edwards, C. A. 1961. La ecología de Symphyla: parte III. factores que controlan la distribución del suelo. Entomologia Experimentalis et Applicata 4: 239-256.
Edwards, C. A. 1990. Symphyla, págs. 891 – 910. En D. L. Dindal [ed .], Guía de biología del suelo. Wiley, Nueva York.
Edwards, C. A., y E. B. Dennis. 1962. El muestreo y extracción de Symphyla del suelo, pp. 300-304. En P. W. Murphy [ed .], Progress in Soil Zoology. Butterworths, Londres.
Eltoum, E. M. A., y R. E. Berry. 1985. Influencia de la lesión de la raíz del sínfilano de jardín (Symphyla: Scutigerellidae) en los procesos fisiológicos en frijoles. Entorno. Entomol. 14: 408-412.
Filinger, G. A. 1928. Observaciones sobre los hábitos y el control del ciempiés de jardín, Scutigerella immaculata, Newport, una plaga en invernaderos. J. Econ. Entomol. 2: 357-360.
Filinger, G. A. 1931. El Jardín Symphylid. Ohio Agr. Exp. Sta. Bul. 486: 1-33.
Getzin, L. W., y C. H. Shanks. 1964. Infección del sínfilo de jardín, Scutigerella immaculata (Newport), por Entomophthora coronata (Constantin) Kevorkian y Metarrhizium anisopliae (Metchnikoff) Sorokin. J. Camino de insectos. 6: 542-543.
Gould, G. E., y C. A. Edwards. 1968. Daño al maíz de campo por sínfilanos. Proc. Indiana Acad. Sci. 77: 214-221.
Howitt, A. J. 1959. Control de Scutigerella immaculata (Newport) en el noroeste del Pacífico con fumigantes de suelo. J. Econ. Entomol. 52: 678-683.
Howitt, A. J., y R. M. Bullock. 1955. Control del ciempiés de jardín. J. Econ. Entomol. 48: 246-250.
Illingworth, J. F. 1927. Sínfidos destructivos para las raíces de la piña. Pineapple News Mar-Dic: 88-91.
Koontz, F. R. 1968. Relaciones biológicas y ecológicas del hongo, Entomophthora coronata (Constantin) Kevorkian, y el sínfilo del jardín,
Scutigerella immaculata (Newport). Ph.D. disertación, Universidad Estatal de Oregón, Corvallis.
Martin, C. H. 1948. Movimiento y poblaciones estacionales del ciempiés de jardín en suelo de invernadero. J. Econ. Entomol. 41: 707-715.
Michelbacher, A. E. 1935. El estado económico del ciempiés de jardín, Scutigerella immaculata (Newp.) en California. J. Econ. Entomol. 28: 1015-1018.
Michelbacher, A. E. 1938. La biología del ciempiés de jardín Scutigerella immaculata. Hilgardia 11: 55-148.
Michelbacher, A. E. 1939. Variación estacional en la distribución de dos especies de Symphyla de California. J. Econ. Entomol 32: 53-57.
Michelbacher, A. E. 1949. La ecología de Symphyla. El Entomol Pan-Pacífico. 25: 1-12.
Peachey, E., A. Moldenke, R. D. William, R. Berry, E. Ingham y E. Groth. 2002. Efecto de los cultivos de cobertura y el sistema de labranza sobre los symphylans (Symphyla: Scutigerella immaculata) y otra biota del suelo en suelos agrícolas. Appl. Suelo Ecol. 21: 59-70.
Riley, H. K. 1929. El ciempiés de invernadero. Indiana Agr. Exp. Sta. Bul. 331: 1-14.
Scheller, U. 1986. Symphyla de Estados Unidos y México. Texas Mem. Mus., Espeleol. Monogr., 1: 87-125.
Sechriest, R. E. 1972. Control del sínfilo de jardín en los campos de maíz de Illinois. J. Econ. Entomol. 65: 599-600.
Shanks, C. H. 1966. Factores que afectan la reproducción del sínfilo del jardín, Scutigerella immaculata. J. Econ. Entomol. 59: 1403-1406.
Simigrai, M. y R.E. Berry. 1974. Resistencia en brócoli al sínfilo de jardín. J. Econ. Entomol. 67: 371-373.
Stimmann, M. W. 1968. Efecto de la temperatura sobre la infección del sínfilan del jardín por Entomophthora coronata. J. Econ. Entomol 61: 1558-1560.
Swenson, K. G. 1965. Infección del sínfilo de jardín, Scutigerella immaculata, con el nematodo DD-136. J. Invertir. Camino. 8: 133-134.
Umble, J. R., y J. R. Fisher. 2003a. Influencia de la alimentación subterránea por symphylans de jardín (Cephalostigmata: Scutigerellidae) en la salud de las plantas. Environmental Entomology 32: 1251-1261.
Umble, J. R., y J. R. Fisher. 2003b. Consideraciones de muestreo para sínfilos de jardín, Scutigerella immaculata Newport, en el oeste de Oregón. Journal of Economic Entomology 96: 969-974.
Umble, J. R., y J. R. Fisher. 2003c. Idoneidad de cultivos y suelos seleccionados para el desarrollo poblacional de symphylan de jardín (Symphyla, Scutigerellidae: Scutigerella immaculata Newport). Journal of Applied Soil Ecology 24: 151-163.
Waterhouse, J. S. 1969. Una evaluación de un nuevo ciempiés predador Lamyctes sp., en el jardín symphylan Scutigerella immaculata. Can. Entomol. 101: 1081-1083.
Waterhouse, J. S., R. Seymour y E. W. Rutkowski. 1969. Efectos biológicos de la inanición en el sínfilo del jardín. J. Econ. Entomol. 62: 338-341.
Woodworth, C. W. 1905. Un nuevo ciempiés de importancia económica. California. J. Tech. 6: 38-42.
Wymore, F. H. 1931. El ciempiés de jardín. Boletín 518 de la Estación Experimental de California: 1-22.
Symphylans: Opciones de manejo de plagas del suelo
Por Jon Umble, Universidad Estatal de Oregón; Rex Dufour, NCAT; Glenn Fisher, Universidad Estatal de Oregón; James Fisher, USDA/ARS; Jim Leap, Universidad de California, Santa Cruz; Mark Van Horn, Universidad de California, Davis
©NCAT
Publicado en febrero de 2006
IP283
Esta publicación es producida por el Centro Nacional de Tecnología Apropiada a través del programa de Agricultura Sostenible de ATTRA, con acuerdo de cooperación con la agencia de Desarrollo Rural del USDA. Esta publicación también fue posible en parte gracias a la financiación de una subvención de Investigación y Educación del Programa de Investigación y Educación sobre Agricultura Sostenible Occidental (Western SARE) del USDA. ATTRA.NCAT.ORG.
Fotos por John Umble, a menos que se indique lo contrario.

