Strawberries: Organic Production

Photo: USDA/ARS
By Martin Guerena, NCAT Agriculture Specialist
Abstract
This publication provides an overview of organic strawberry production methods. It also covers integrated pest management (IPM) and weed-control techniques that can reduce pesticide use in strawberry production. It includes discussions of weeds, pests, diseases, greenhouse production, plasticulture, fertility, economics, and marketing. It also provides further resources.
Contents
Introduction
Planting Systems
Varieties
Fertility
Weed Control
Insect and Mite Control
Disease Control
Greenhouse Production
Tunnel Production
Harvest and Postharvest
Economics
Marketing
References
Further Resources
Introduction
Strawberries are a viable crop in most areas of the United States. Cultivars have been developed to suit most climatic conditions. In many locations, demand for locally produced berries far exceeds available supplies; small-scale producers can thus get higher returns from strawberries than from most other crops. However, it is important to consider the cost of production to ensure you are making money because the costs of organic strawberry production can be considerable.
Organically grown berries may command a price premium. Organic production excludes the use of synthetic fertilizers and conventional pesticides, and requires soil building and integrated pest controls. Federal organic standards restrict claims of “organically grown” to those farms that are certified to be organic by a USDA-accredited certification agency. For more information, check ATTRA’s Organic Farming page. Excellent cultural information for conventional strawberry production—planting systems, pest control, cultivar recommendations, etc.—can be obtained from the Cooperative Extension Service in most states. We have not attempted to develop a one-size-fits-all prescription for organic (or other ecologically based) strawberry production. Rather, we have introduced the most common challenges and offered some possible solutions and factors for consideration.
For many years, until it was phased out in 2017, conventional strawberry growers routinely used the soil fumigant methyl bromide to control weeds, soilborne diseases, nematodes, and soil-dwelling insects. Now chloropicrin, Telone, and other minor fumigants are used, but these are being scrutinized because of the need for increasing buffer zones near homes, schools, and communities, in addition to required worker protections (Guthman, 2017). There are feasible alternatives in strawberry production, as many organic growers can attest.
Planting Systems
Planting systems for strawberries vary, depending on the environment and production goals. The grower must decide the relative priorities of yield, size, flavor, or other qualities of the fruit, and seek a system that balances these goals. Systems that focus primarily on yield are the least sustainable because of the enormous amount of energy used for maintenance, plastic, and transportation. In many of these systems, the plants are grown on raised beds as annuals. This results in removal of the plants, plastic mulch, and irrigation system at the end of every season. Regardless of the system used, conventional yields are usually higher than organic yields. However, prices and profits for organic strawberries can be more than $12,000 per acre higher in California (Bolda et al., 2014).
Raised Bed Plasticulture
Organic and conventional growers in California, Florida, and parts of the Southeast, where most of the nation’s strawberries are produced, tend to favor raised-bed plasticulture. They grow plants as annuals, transplanting strawberry crowns in the late summer or early fall. Production starts in the late winter and continues through the summer and into late fall, depending on the area and the varieties grown. Because methyl bromide is not allowed in organic production, solarization, biosolarization, crop rotation, green manure crops, and compost are critical to control soilborne diseases and pests. Two types of raised beds are used in these intensive systems. Narrow beds have two rows of plants with one drip line running between them. The distance between beds averages 40 inches. Drip tape is buried at a depth of about 2.5 inches. Wide beds usually have four rows of plants and two drip lines, with 64 inches between beds. Spacing between plants in both types of bed averages 12 to 14 inches.

Strawberries in a basket. Photo: USDA/ARS
Plastic mulch is used in both narrow and wide beds and can vary from a single strip of plastic laid between the plants to full bed coverage, where holes must be punched for the plant to develop. Some conventional growers in California use clear plastic, which warms the bed faster, stimulating early season growth; these growers use fumigation to control most weeds. Black plastic is used in organic production, primarily for weed control. Because the black plastic prevents the sun’s rays from penetrating, the beds remain cool, resulting in slower initial growth of the plants and reduced irrigation frequency compared to clear plastic mulch. There is a plastic mulch on the market that selectively permits soil-warming radiation to penetrate while eliminating the light that promotes weed growth. This type of plastic is preferred by growers in the Southeast.
Raised beds provide good drainage. Because they make the flowers and fruit easier to see and reach, raised beds also help growers forecast yields, while making harvesting easier and faster. Some growers dig deep furrows between the beds so that harvesters do not have to stoop so low to search for fruit. In cold climates, plants in raised beds may be prone to freeze damage. Still, raised beds usually out-produce flat beds. Due to increased aeration and protection from splashing soil particles, plants in plastic-mulched raised beds have less disease. At the end of the season, some growers pull the strawberry plants out and keep the beds to transplant summer/fall vegetables. Machinery is available to shape the bed, lay out the irrigation line, and cover the bed with plastic mulch, all in a single pass.
Plasticulture, Sustainability, and Organic Farming
Plasticulture is not without its serious critics. The plastic has to come from somewhere, and it has to be discarded at the end of the one- to three-year production cycle. Clearly, critics say, this is not an environmentally sustainable system. And, says Cornell University fruit researcher Marvin Pritts, PhD, if you consider all the environmental costs to society, plasticulture is also not economically sustainable in the long run. Pritts also points out that even more plastic—in the form of row covers, tunnels, hoop houses, etc.—is needed to make the system work in cold climates.USDA researchers have shown that fields mulched with plastic cause four times more water runoff than fields mulched with organic materials. Due to this high rate of runoff, fields mulched with plastic suffer up to 15 times more soil erosion than fields mulched with organic matter (Anon., 1999). However, planting grasses or other types of vegetation alongside drainage ditches can reduce the rate of erosion and provide habitat for beneficial insects.
Yet, even organic growers—especially those in California, where plasticulture has reigned the longest—are buying into the plasticulture production model. Why? The answer is weeds. Strawberries are notoriously prone to weed encroachment with resultant loss of productivity. Plasticulture provides good to excellent weed control without herbicides. The USDA National Organic Program (NOP) states that plastic or other synthetic mulches are allowed in organic production, provided that they are removed from the field at the end of the growing or harvest season.
Pritts admits that implementing alternatives to plasticulture requires well-informed and committed management. Moreover, production in each locale may require fine-tuning to get the right mix of groundcovers and timing for planting, mowing, and other manipulations. This is probably not going to be as easy as rolling out the plastic sheeting. Nevertheless, using small amounts of post-emergent herbicide, though not allowed in organic production, may be more sustainable than the continued use of tons upon tons of non-renewable, non-recyclable plastic mulch. There are “biodegradable” plastics on the market made from plant starch, but, due to its brittleness, the plant starch must be blended with petroleum-based polymers, which are considered synthetic materials by the NOP standards. For more information on biodegradable plastic, check Current and Future Prospects for Biodegradable Plastic Mulch in Certified Organic Production Systems in the Further Resources section.
Matted Row System
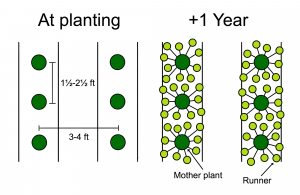
Matted row strawberry system. Photo: Garlan Miles, wikimedia.org
In this system, the crowns are planted in early spring. As the plants produce flowers, the blossoms are removed to encourage runner (or daughter plant) production. The daughters root on the bed and produce a crop the following spring. Weeds can be a problem in this system, and dead leaves and other debris must be removed to reduce disease and pest problems. However, once established, this system can produce for three to four years, depending on pest pressure. The distance between plants is 18 to 24 inches, and the distance between the rows varies from 36 to 50 inches, depending on the cultivation equipment used. According to Marvin Pritts of Cornell University, the matted row system offers northern strawberry growers a low-risk system that requires less focus and time than annual plasticulture systems (Pritts, 2002). This system is better suited for the small-scale, home-gardener producer in colder regions because costs are lower, it maximizes garden space, and it takes advantage of the natural growing habit of the strawberry plant.
Ribbon Row System
This system can employ high-density or low-density planting on a single row. With low-density planting, the spacing is 12 to 36 inches between rows and 14 to 18 inches between plants. With high-density planting, the distance between the rows is the same, but the distance between plants varies between four and 12 inches. The crowns are planted in the fall. Once they start blooming, the flowers are not removed, and fruit is produced in the first season. Runners are removed to stimulate flower formation and to increase fruit size. At the end of the second season, the planting can be changed to the matted row system by letting the runners fill in empty spaces on the beds.
Varieties
Selection of appropriate varieties is important. Besides determining yields and quality, the variety also determines production seasons and pest- control practices. Your county Extension agent can usually recommend varieties that have been shown to respond well to the area’s climatic conditions.
However, variety trials are usually conducted utilizing conventional production systems. The variety’s performance may be different in an organic system. Therefore, organic growers are advised to plant more than one of the recommended varieties and conduct their own variety trials. Other organic growers in your area may also be able to advise you.
Strawberry varieties are classified as either “June-bearing” or “day-neutral” varieties. June-bearing, or short-day, varieties start forming flower buds as the day length gets shorter and temperatures get cooler and generally produce one large harvest in late spring or early summer. The following are examples of June-bearing varieties: Allstar, Annapolis, Benicia, Benton, Camarosa, Camino Real, Cavendish, Chandler, Earliglow, Firecracker, Gaviota, Glooscap, Honeyoye, Hood, Jewel, Kent, Mesabi, Mojave, Northeaster, Shuksan, Strawberry Festival, Surecrop, Sweet Charlie, Tillamook, and Todem.
Day-neutral varieties are insensitive to day length and produce two to three fruit crops over the course of the summer and into the fall, with the larger crop coming in the summer, as long as night-time temperatures drop below 60° F (Strand, 1993). Day-neutral varieties include the following: Albion, Alinta, Aromas, Cabrillo, Diamante, Evie 2, Hecker, Monterey, Portola, San Andeas, Seascape, Selva, Sweet Ann, Tribute, and Tristar.
Fertility
There are at least two aspects of strawberry production that are unique and distinct from other perennial fruits with respect to fertility. June-bearing strawberries set buds for the following year’s fruit in the fall. (Most perennial fruit crops set their fruit buds in the spring or early summer.) To get a good bud set, the plants must have adequate chilling and not be nutritionally stressed. Therefore, fertilizer applications are usually warranted in the late summer, giving the organic fertilizer material enough time to break down and provide nutrients for the plants during the crucial fall bud-set.
Timing is critical in supplying nitrogen to berry crops, and the nitrogen-release rates for organic fertilizers may not match the nitrogen needs of the crop. A study on organic fertilizers in California found great variability in the nitrogen availability of different sources of fertilizers (Gaskell, 2004). These included guano, feather meal, liquid fish emulsion, fish meal, pelleted chicken manure, compost, and a green manure crop. Initially, the soil nitrate nitrogen from the green-manure crop and compost kept the level of nitrogen at adequate amounts (50 to 75 ppm) for three to four weeks and then declined to background soil levels below 10 ppm. Supplemental fertilizing is therefore necessary to carry the crop through the season. Strawberry producers using the annual plasticulture system must rely on soluble organic fertilizers applied through drip irrigation lines. Farmers using these systems must face solubility issues and consider the capacity of these products to be filtered through fine mesh without plugging drip emitters. Products injected into the system may not emerge at the same concentration. In other systems, foliar or side-dress applications will be warranted.
Although all perennial fruit crops will benefit from the fertility provided by pre-plant cover-cropping and green-manuring, strawberries are so prone to weed problems that pre-plant preparations to reduce weed pressure are practically mandatory in organic production. A thick cover crop of a grass/legume mix will help to smother out many weeds and will provide important long-term improvements in soil fertility and soil organic matter. In areas such as coastal California, long growing seasons and high land rents may make the extended use of cover crops uneconomical. However, many growers believe that the long-term benefits of cover crops and rotations to soil fertility and pest and disease suppression are worth the cost. Compost can be used as a supplement or alternative. Spreading and incorporating the compost on the beds only, avoiding the furrows, will help concentrate fertility and microorganisms where they are most needed. Compost application rates vary from 3 tons/acre to 10 tons/acre. Supplemental fertigation is necessary to carry the plants through the production season.
Research from Ohio has shown that vermicompost (compost made from earthworm waste) applications increased strawberry growth and yields significantly (Arancon et al., 2004). These responses seemed not to be dose-dependent. Strawberries at one site grew fastest and yielded most in response to the 10 ton/hectare (4.05 ton/acre) vermicompost application rate, whereas strawberries responded positively and similarly to both the 5 ton/hectare (2.02 ton/acre) and 10 ton/hectare rates of application at another site. These responses could not have been mediated by the availability of macronutrients, since all plots were supplemented with inorganic fertilizers to equalize macronutrient inputs for all treatments. Based on other research in the laboratory, however, the responses could have been due to production of plant growth regulators by microorganisms during vermicomposting.
Foliar application of aerobically-prepared compost tea increased yields in a British Columbia, Canada, study (Welke, 2004). Besides reducing incidences of Botrytis, the compost-tea treatment increased yields in strawberries by 20% compared to the control and water sprays.
For more information on organic fertilizers, vermicomposting, compost, and cover crops, look for these ATTRA publications: Composting: The Basics, Alternative Soil Amendments, and Overview of Cover Crops and Green Manures.
Weed Control
Weeds are one of the biggest problems that organic strawberry growers face. Pre-plant site preparation is critical. Refer to ATTRA’s tipsheet Tree Fruits: Organic Production Overview for site-preparation strategies, as well as for basic weed-control ideas.
Cultural Methods
Organic growers will find that some hand weeding is necessary. Weeds in organic plasticulture systems can become troublesome even where black plastic mulch is used. In such situations, the weeds emerge from the planting holes made for the strawberry plants. The rows must be straight, and the plastic laid precisely, to allow mechanical cultivation of the furrows without damaging the beds and plastic.
A variety of colored mulches have been studied in California to determine their contribution to weed control and crop response. Black mulch provides the best weed control but does not warm the soil as well as clear plastic. Soil warming with clear plastic results in plants that grow and produce earlier in the season, but weeds are not controlled. Research determined that the effect of mulch color on transmittance of photosynthetically-active light (400 to 700 mm) through mulches was the key weed-control factor (Johnson and Fenimore, 2005). Green and brown plastic mulches provided the best combination of soil warming and weed-control benefits at all trial locations.
Anaerobic Soil Disinfestation (ASD) is a process that occurs in tarped, saturated soils as microorganisms deplete oxygen and start consuming carbon, which in turn produces toxins such as fatty acids (acetic, propionic, butyric, valic, and caproic acids), as well as aldehydes, alcohols, ammonia, metal ions, and other volatile organic compounds (Gamliel and Stapleton, 1997). This biological fumigation can suppress various pests in the soil, like diseases, nematodes, and weeds. Plastic used in ASD can be clear or black. When clear plastic is used, the process is known as biosolarization and can enhance efficacy and reduce treatment period. Dark plastic is effective but can take three to five weeks to work, depending on the climate and soil temperatures (Testen and Miller, 2017). See box on page 11 for more information on ASD, solarization, and biosolarization.
Occultation is a weed-control method that consists of covering the soil with a black opaque plastic sheeting for four to eight weeks before planting. Having the soil moist under the plastic, combined with the heat generated by the black surface, creates conditions to start germination of weed seeds, which are then blocked in their development by the absence of light. Once the plant spends its energy reserves searching for light, it withers and dies.
The matted row system (where plants from runners form a 6- to 30-inch-wide solid bed) is commonly used by strawberry growers in many regions of the United States. This method precludes mechanical cultivation for weed control within the bed, though cultivation is commonly used to renovate or narrow a bed. Weed problems tend to increase with the age of the planting. Many organic growers have therefore chosen shorter fruiting rotations. That is, a bed may be allowed to fruit for two seasons before it is turned under and replanted to a cover crop.
Planters’ paper, a black paper mulch, was used in matted rows for a biodegradable mulch study (Weber, 2003). It reduced weeds but degraded quickly along the edges where it was covered by soil, allowing the wind to tear and blow large pieces off the plots. The rate of degradation the first year was quick, but the paper still reduced weed population compared to clear mulch and the control.
Mechanical Methods
In plasticulture systems, harvest crews are sometimes used to weed when the weed pressure is high or when the harvest day is short. Weeds will grow through the plant hole and need to be removed by hand when the berry plants are small. Mechanical cultivation can be done to address the weeds between the beds, in the furrows.
Biological Methods
Before the widespread adoption of herbicides, geese were commonly used for weed control in commercial-scale strawberry production. In areas of concentrated crop production, farmers often had the benefit of weeder-geese services for hire. Weeder geese can still be used to control grasses and a few broadleaf weeds, but close management of the geese is essential. In any case, the geese must be removed before fruiting season, because they will eat strawberries before going after grass. Under the USDA National Organic Program (NOP) organic standards, raw animal manure must be composted unless it is incorporated into the soil not less than 120 days prior to harvest of a crop whose edible portion has direct contact with the soil surface or soil particles. Therefore, geese would need to be removed from the field and their manure incorporated at least four months prior to the beginning of strawberry harvest. Other animals used for weed control on strawberries include sheep and guinea fowl.
Other forms of biological weed control usually involve the use of insects or pathogens. They are weed-specific and will not attack other weeds or the crop plants. It is unlikely that biological controls will completely eradicate weed species associated with strawberry production.
Organic Mulches
Strawberry plants, especially in the North, are commonly mulched with straw over the winter to minimize cold damage. In the spring, the straw is raked into the aisles where it provides some control of weeds and helps to keep the berries clean. Caution must be taken with some organic mulches, in that they may harbor pests like snails, slugs, cutworms, earwigs, and sow bugs. On the other hand, straw provides excellent habitat for spiders and has been known to reduce diseases. A study in Ohio showed that straw mulch between strawberry rows was equal to or more effective than fungicides for controlling leather rot (Phytophthora cactorum) (Ellis et al., 1998).
Newspaper can make a good mulch for gardeners and smaller operations. Apply it on the beds with enough sheets to have around a 1-inch layer, then poke holes and plant the crowns into the mulched beds. Or, plant the crowns then place the sheets of newspaper around the plant, leaving about three inches around the stem. Spray the newspaper with water to dampen it and settle the mulch to the bed. Place additional layers of organic matter, such as straw, grass clippings or leaves, to weigh down the paper and provide an extra layer of protection against weeds emerging through. Only newspaper or other recycled paper, without glossy or colored inks, may be used as mulch under the NOP standards.
Woolen landscaping fabric was the best alternative treatment in a study conducted in Minnesota (Forcella et al., 2003). A one-ply woolen fabric centered over the crop nearly eliminated weeds from rows, promoted daughter-plant rooting, and allowed maximum fruit yield, equivalent to that obtained in plots that were hand-weeded.
Thermal Controls
Thermal technology, from flamers to infrared burners, keeps evolving with new products emerging onto the market. At present, thermal control methods include handheld flamers, mounted row crop flamers, infrared weeders, steamers, hot water, and hot foam. Timing is critical for successful thermal control of weeds. The younger the weed, the easier it is to desiccate. Grasses can be burned back, but the growing point usually sends out new growth. Some of these devices may not fit in a particular system, but others may be successful components of a weed-control program.
Organic Herbicides
These products are effective in controlling weeds when the weeds are small and the environmental conditions are optimum. Broadleaf weeds at the cotyledon and first-true-leaf stage are easier to control than older plants. Grasses will burn back but grow again, due to the growing point being below the leaf surface. Organic herbicides only kill contacted tissue; thus, good coverage is essential. Perennial weeds will have leaves burned off but will recover and continue growing, so organic herbicides are not recommended if you have perennial weed pressure.
Woven Synthetic Fabric Mulches
Synthetic fabric mulches (trade names: Weed Lock, Weed Barrier, Weed Stopper, etc.) offer some of the same weed suppression as regular plastic mulches, but have the advantage of being water- and air-permeable. Though initially more expensive than regular plastic, the higher-quality grades of fabric mulch can be used year after year. These woven mulches are used in essentially the same way as plastics in the systems described above. However, because they are water-permeable, it should not be necessary to add irrigation lines under the mulches in areas with adequate rainfall.
Insect and Mite Control
Numerous insects feed on strawberry plants and threaten yields. Your county Extension service will be familiar with pests common to specific areas and can help with proper identification, which is the first step in pest management. A scouting program with regular monitoring can help growers determine both the pest pressure and presence of beneficial insects. Once pest pressure reaches the economic threshold, control actions are necessary. If biological controls are to be used, they must be deployed before the pests reach critical levels. That is why monitoring is so important. In large operations, where harvest crews are used regularly, training the crew foreman to identify insect pests and diseases can help in the monitoring process.
Beneficial-insect habitats planted alongside strawberry fields provide shelter, pollen, and nectar sources to predators and parasites of insect pests. Beneficial insects are able to take refuge in the habitat when fields are treated with a pesticide. When purchased beneficial insects are released, these habitats encourage the beneficials to remain and continue their lifecycles, helping reduce pest populations. Some pests may also inhabit the refuge along with beneficials, so it is important to monitor these habitats. For additional information, request ATTRA’s Biointensive Integrated Pest Management, Farmscaping to Enhance Biological Control, and A Pictorial Guide to Hedgerow Plants for Beneficial Insects.
White Grubs
Primarily a problem in the eastern United States, white grubs can cause serious damage if strawberries are planted immediately after a sod crop. White grubs are the larvae of May and June beetles and other beetles in Scarabaeidae. Late-summer or early-fall plowing destroys many larvae, pupae, and adults in the soil and also exposes these stages to predators. The milky-spore-disease bacteria, Bacillus popillae and Bacillus lentimorbus, are important natural enemies of Scarab beetles. Grubs ingest spores of these bacteria on the thatch or roots of the grasses they eat. The spores then germinate and the bacteria multiply inside the grubs, which die and disintegrate, leaving many new, viable spores to spread the disease to succeeding generations (Daar, 1988).
Beneficial nematodes are also effective against soil-dwelling grubs. Steinernema carpocapsae will infect its host near the soil surface, while Heterorhabditus bacteriophora actively searches for its host below the soil surface (Flint and Dreistadt, 1998). These nematodes and milky-spore bacteria are widely available through mail-order garden supply companies.
Strawberry Clipper
(Strawberry Bud Weevil)
The strawberry clipper or bud weevil, Anthonomus signatus, occurs only east of the Rockies. Adult beetles emerge in the early spring and lay eggs in the buds, girdling them and preventing them from opening, then they use their snouts to pierce the flower buds and feed on pollen. Contrasting studies on strawberry clipper have been conducted. One study found that most of the 12 varieties studied compensated for a significant amount of flower bud loss, provided that the loss occurred early in the development of the inflorescence (Pritts et al., 1999). A later study showed that liberal thresholds developed from the previous studies were exceeded in two of the three research sites and damage levels were severe enough to reduce yields significantly (Handley et al., 2002). The clipper moves at the very slow rate of 30 feet per season. In a new planting, it is unlikely that the damage would extend more than 30 feet from the perimeter into the plot. Damage may be somewhat more extensive in older plantings, but still limited by the rate of movement of the clippers (they will have moved approximately 60 feet into a two-year planting and 90 feet into a three-year planting). Organic growers should destroy damaged buds, which contain eggs, eliminate trash and nearby foliage that provide hibernation sites for adult weevils, and apply an organically approved insecticide as a last resort.
Strawberry Rootworm
Strawberry rootworm (Paria fragariae) adults feed mainly at night, making holes in the leaves. The larvae feed on fine roots and eat the crowns close to the ground. Cultural control consists of plowing infested fields after harvest, crop rotation, and setting new plantings away from woods (favorable hibernation sites) and from older strawberry plantings. Apparently, IPM damage thresholds have not been established for the rootworm. If the grower feels that pesticide treatment is necessary based on scouting, nocturnal treatment should be aimed at the foliar-feeding adults with an organically approved pyrethrum-based insecticide. Soil-dwelling predators such as ground beetles or insect-attacking nematodes like Steinernema species may provide some control.
Strawberry Root Weevil
The adults of these species feed mostly on leaves, causing minor damage. The larval stage is the problem, as the larvae feed on roots and crowns of the strawberry plants. Root weevils have many alternate hosts, including other small fruits, cranberries, grapes, mint, hops, and many ornamental plants. Rotation with non-host crops like corn, wheat, clover, and alfalfa can reduce populations (Berry, 1998). Like other ground-dwelling pests, strawberry root weevils are susceptible to attack from ground beetles and from parasitic nematodes such as Steinernema or Heterorhabditus species. The root weevils are crawling insects that also have been excluded from fields by fences, trenches, and barriers like sticky tape (Bomford and Vernon, 2005; Strand, 1993). Adult controls are similar to the strawberry rootworm: nocturnal treatment with an organically approved pyrethrum-based insecticide.

Lygus bug. Photo: USDA/ARS
Lygus Bugs
The tarnished plant bug or lygus bug (primarily Lygus lineolaris in the East and L. hesperus in the West) can be troublesome, especially in plantings of day-neutral varieties, which fruit throughout the growing season. Adults and nymphs (the nymphs cause the most damage) suck sap from the plant and inject a toxic saliva. This feeding results in a characteristic deformation of the fruits called cat-facing, which makes the berries unusable and unmarketable.
Keeping any groundcover well clipped for a distance of five to 10 yards around a strawberry field, and otherwise destroying places favorable for hibernation, may help reduce lygus bug populations. Adult lygus bugs hibernate under leaves, stones, and bark. They usually lay eggs in the stems of herbaceous cultivated plants and broadleaf weeds. Legumes (vetches, clovers, alfalfa, etc.) can harbor large populations of these pests. This must be considered if beneficial habitats using these plants are established near strawberry plantings.
Trap crops are also useful in lygus bug management. In California, an annual trap-crop mix of one dormant and one semi-dormant alfalfa variety, two radish varieties (Daikon and Cherry Belle), and sweet alyssum has been used with success. Lygus bugs move in from surrounding fields and settle on the trap crops, which can then be treated with insecticides or vacuumed (Dufour, 2000). Bug vacs range from tractor-mounted machines to small hand-held devices and are actually vacuum cleaners for pests. Research done in Watsonville, California, demonstrated that lygus bugs were more attracted to a field-edge alfalfa trap crop than to a radish/mustard or strawberry row (Swezey, 2004). Vacuuming the alfalfa trap crop with a tractor-mounted bug vac reduced damage due to lygus bug feeding in associated strawberry rows, when compared to vacuuming the whole field.

Bug vacuum. Photo: USDA/ARS
This saved operating costs of the bug vac and increased marketable fruit.
Another study using alfalfa as a trap crop in organic strawberries found that insect and spider predators of lygus aggregated in the alfalfa and had a significantly higher proportion of their population contain Lygus spp. remains than those predators collected from nearby rows of strawberries. These results suggest that alfalfa trap cropping might be a useful tactic for conserving the biological control services of generalist predators in organically grown strawberries in California (Hagler et al., 2018).
Research conducted in New England found variation in susceptibility to the lygus bug among 20 strawberry cultivars (Handley et al., 1991). Honeoye, Sparkle, Veestar, and Canoga suffered the least from feeding, while Kent, MicMac, Scott, Blomidon, and Redchief suffered most. A fungus, Beauveria bassiana, has some efficacy against lygus bugs. In New York, three years of tests concluded that the commercial formulation of B. bassiana, Mycotrol™, reduced lygus damage about 50%, compared to untreated controls, but was still considerably less effective than synthetic insecticides such as malathion (Kovach and English-Loeb, 1997). Mycotrol worked best when targeted at younger nymphs and when humidity levels were adequate. In combination with other cultural controls (choosing the right cultivar and close mowing near the planting), use of Mycotrol O Botanagard or Naturalis could be of help to organic growers in controlling lygus.

Parasitic wasp Peristenus digoneutis. Photo: USDA/ARS
Although the lygus bug has several natural insect enemies, none of the native ones have proved consistently effective in providing a commercial level of control in strawberries. In California, Peristenus digoneutis and P. stygicus were released in 1998. They have become established and annual increases in parasitism were noted in 2000-2002 (Fuester et al., 2004). Higher rates of parasitism by P. digoneutis were observed in New York on organic or casually sprayed farms than on intensively treated farms (Tilmon and Hoffmann, 2003). Anaphes ioles is a lygus egg parasitoid that has been used in California and in other states with some success. Researchers who released 15,000 A.ioles weekly on one-acre strawberry plots observed a 64% suppression of Lygus hesperus, compared to a 44.7% reduction achieved with a pesticide application (Udayagiri et al., 2000).
Since lygus nymphs are most troublesome, aim scouting efforts at this life stage. Start checking for nymphs as soon as flowers appear. Tap 10 to 15 flower clusters over a white plastic saucer, so that the bright green nymphs can be seen and counted. Determine the average number of nymphs per cluster (total number of nymphs divided by total number of clusters). If sampling is concentrated near weedy borders, the action threshold is one nymph per cluster, but if done randomly throughout the planting, 0.5 nymphs per cluster should be considered adequate to prompt a pesticide treatment (Kovach et al., 1993). However, Cornell researchers caution that growers who intend to use the slow-acting biological insecticide B. bassiana may need to use a lower threshold (Kovach and English-Loeb, 1997). If other natural enemies of lygus are present—such as spiders, bigeyed bugs (Geocoris species), assassin bugs (Zelus and Sinea species), damsel bugs (Nabis species), and lacewing larvae (Chrysoperla species)—you might want to consider adjusting the threshold numbers accordingly.
Mites
The web-spinning spider mites are in the genus Tetranychus, which includes the two-spotted spider mite, Pacific spider mite, and strawberry spider mite, among others. These plant-feeding mites consume juices from strawberry leaves. Large populations can reduce photosynthetic capacity, resulting in weakened plants and reduced fruit yields. Some growers who do not often use pesticides may see very few mites; the natural enemies of the mites will usually keep them in check. These natural enemies include other mites, such as Phytoseiulus persimilis, Metaseiulus occidentalis, and Neoseiulus californicus, and insects like bigeyed bugs, damsel bugs, minute pirate bugs, lacewings, spider mite destroyers, and sixspotted thrips. Growers can buy some of these predators from commercial insectaries to release on the farm. The predators can also be attracted and conserved naturally through the use of insect habitats. Insecticidal soaps, “narrow range” oils, vegetable oils, neem-based products such as Trilogy®, and sulfur are acceptable miticides in organic production (check with your certifier regarding specific products). Application must thoroughly cover the leaves’ undersides, and products that are diluted must be applied in high volumes (more than 100 gallons of water per acre) to achieve complete coverage. Both oils and soaps can burn plants if over-applied or if high temperatures (greater than 80° F) occur during and after treatments. Metarhizium brunneum, a commercially available insect pathogenic fungus, was found to have systemic properties in strawberry plants and appeared to withstand spider mite infestations better than untreated controls (Dara and Dara, 2015).
A scouting method to determine if spider mites are at a level requiring treatment is as follows: select a random sample of 10 leaflets per acre and if mites are present at densities approaching five per leaflet, then a treatment may be in order (Burrack, 2017). Again, the number of natural enemies should also be considered when determining a threshold for chemical treatment. While this scouting method is probably applicable to most areas, growers should check with their local Cooperative Extension Service for scouting guidelines.
Contact Information for Beneficial Organism Suppliers
Companies that sell mites and other beneficial organisms are listed in the University of Kentucky’s Vendors of Beneficial Organisms in North America. This online PDF publication contains contact information for major commercial suppliers of the more than 100 beneficial organisms that are currently used in biological pest control. It not only indexes the suppliers by the natural enemies they sell, but also matches the beneficial organisms with their target pests.
Some of the mites you see when scouting may be predator mites. You may need a magnifying glass to distinguish between these beneficial mites and the pest mites. One key to telling them apart is that the beneficial predator mites are generally more active than the two-spotted mites—they typically move quickly about the leaf surface looking for prey. Depending on your geographical area and the species involved, the recommended ratio of beneficial mites to pest mites varies, but seems to average approximately 1:10. That is, if there appears to be at least one beneficial mite for every 10 pest mites, control of the pest mites will probably be achieved naturally without the intervention of miticidal sprays.
Dust that accumulates on the spider mite’s webbing creates an ideal shelter for the mites and their eggs. These little dust “tents” discourage predators and prevent the miticide from reaching the mites and their offspring. California growers commonly water roads, post “slow” signs, plant windbreaks and beneficial insect habitats, and use fencing to decrease dust in strawberry fields. Other cultural practices include use of tolerant cultivars, precise chilling that depends on the cultivar, nursery management, and supplemental pre-plant cold storage.
Other Pests
Other arthropods that will occasionally reach pest status include aphids, spittlebugs, whiteflies, Cyclamen mites, various caterpillars, earwigs, and leafhoppers. If these become a problem, consult your local farm advisor, visit the numerous websites listed in Further Resources, or contact ATTRA.
Disease Control
Diseases in plants occur when a pathogen is present, the host is susceptible, and the environment is favorable for the disease to develop. Altering any one of these three factors may prevent the disease from occurring. Organisms responsible for plant diseases include fungi, bacteria, nematodes, and viruses. If these organisms are present, then manipulation of the environment and the host, to make it less susceptible, help manage diseases on all crops, strawberries included. Soil health and management are the keys for successful control of plant disease. A soil with adequate organic matter can house numerous organisms, such as bacteria, fungi, nematodes, protozoa, arthropods, and earthworms, that may suppress soil-borne pathogens. This disease suppression is caused by either antagonism, competition for nutrients, or competition for space around the root (the rhizosphere) and induced systemic resistance (ISR) or systemic acquired resistance (SAR) triggered in the plant. Increasing soil organic matter by incorporating cover crops or adding compost and organic fertilizers will help maintain these beneficial organisms. For more information, see the ATTRA publication Sustainable Management of Soil-Borne Plant Diseases.
Rotating strawberries with other crops is a critical factor in organic production, and many certifying agencies require it as a component of the Organic System Plan. Crop rotation reduces insect, disease, and weed pests, improves soil fertility, improves soil tilth and structure, reduces soil erosion, and improves water management. Cover crops, vegetable crops, legumes, and cereals are recommended rotation choices. Avoid solanaceous crops like tomatoes, potatoes, peppers, and eggplant that may harbor diseases such as Verticillium. Research in the Salinas Valley of California found that incorporating broccoli residues reduced Verticillium dahliae in the soil and that rotation with broccoli may be a feasible approach to manage Verticillium diseases in susceptible crops (Subbarao et al., 1999).
Compost teas or extracts and other innovative concoctions such as yeast-sugar solutions, sodium bicarbonate (baking soda), and milk have become popular as foliar disease preventatives among many organic growers. Compost teas and yeasts introduce non-plant-pathogenic microorganisms and biocontrol agents that may compete with and antagonize disease spores as they try to establish themselves on the host. Baking soda works at the chemical level, interfering in spore germination.
Elemental copper and sulfur have long been used by conventional and organic growers as pesticides for foliar bacterial diseases and powdery mildew, respectively.
Root Rot Complex
Soil-borne fungi such as Phytophthora, Pythium, Rhizoctonia species, and Verticillium dahliae are major pathogens that affect strawberries worldwide. In organic production, the cultural methods described above—crop rotation, compost application, and solarization/biosolarization—aid in the control of these diseases. Other cultural controls include using resistant varieties, planting strawberries in a pathogen-free, well-drained soil, avoiding over-watering, and planting only certified disease-free plants. Some growers inoculate the soil or the plants with a variety of commercially available biological products, such as Vesicular Arbuscular Mycorrhizae (VAM) or Arbuscular Mycorrhizae (AM), Trichoderma species (Rootshield, Promot, SoilGard), Streptomyces griseovirdis (Mycostop), and Streptomyces lydicus (Actinovate).
Soil Solarization, Anaerobic Soil Disinfestation (ASD), and Biosolarization
Imagine harnessing the sun’s energy to destroy your enemies. Like Archimedes—the ancient Greek who used mirrors to concentrate sunlight to burn the Roman fleet—farmers can destroy or disable insects, diseases, nematodes, and weeds in the field. The technique known as solarization consists of laying clear plastic mulch on moist soil. Heat is trapped under the plastic, raising the soil’s temperature and killing or debilitating pests. Usually, this soil pasteurization process takes four to six weeks, but the amount of time depends on many factors, such as rain, wind, day length, soil texture, and the quality of the polyethylene mulch. Ultraviolet-protected plastic makes it possible to remove and re-use the mulch.Anaerobic soil disinfestation (ASD) is a biological process that occurs when saturated soils are tarped with an incorporated organic-matter source. Microorganisms deplete the oxygen and convert carbon into fumigant-like toxins. The process of ASD for strawberry production is as follows:
- Broadcast organic matter (compost, green waste, or pomace) or disk down a cover crop.
- Incorporate the organic matter into the soil.
- Form beds and lay down drip tape.
- Cover the beds with plastic mulch.
- Irrigate using the drip tape until soil is saturated.
- Wait two to six weeks (depends on climate, soil type, and if irrigation needs to be repeated to achieve anaerobic conditions).
- Punch holes in the plastic, let it air out, then plant.
Biosolarization combines solarization with anaerobic soil disinfestation (ASD), which only takes one week, compared to the four to eight weeks for solarization and two to six weeks for ASD with black plastic. ASD creates temporary anaerobic conditions in the soil that encourage anaerobic microorganisms that break down available carbon sources, producing organic acids, aldehydes, alcohols, ammonia, metal ions, and volatile organic compounds that are toxic or suppressive to soil pests and diseases (Momma, 2008; Dominguez et al., 2014; Huang et al., 2015; van Agtmaal et al., 2015; Achmon et al., 2017). Research conducted in Spain and California has demonstrated that solarization has potential as a component in an IPM program for root diseases in strawberry production (Hartz et al., 1993; Pinkerton et al., 2002; Dominguez et al., 2014). For more information, check the ATTRA Soil Solarization and Biosolarization Tipsheet.
Anthracnose
Anthracnose can be very serious, causing strawberry plants to die out in midsummer. The disease produces a rust color throughout the crown and eventually stops the plants from growing. Symptoms are most noticeable during summer dry spells. Because high soil fertility favors anthracnose, little or no fertilizer should be applied when disease pressure is strong. However, resistant cultivars (Flavorfest, Sweet Charlie, and Winterstar) can be grown successfully at much higher fertility levels (Maas, 1987). Anthracnose is more prevalent in the Southeast than elsewhere. Commercial growers in the Southeast should avoid planting on former strawberry sites and use locally adapted resistant cultivars. Additionally, using low or high tunnels covers the plant, reducing fungal spores to decrease or eliminate anthracnose incidence (Demchak et al., 2019).
Botrytis (gray mold)
Gray mold, caused by the fungus Botrytis cinerea, is one of the most common and serious fruit rot diseases. The fungus grows best in cool, damp weather, and gray mold can be devastating if rainy weather coincides with harvest, when strawberry fruit is at its ripest and most susceptible. Pickers handling infected berries can spread the infection to healthy berries. Control of gray mold is aided by removing infected debris from the field and by providing good drainage. Infected fruit can be picked off the plants and placed in the furrow, as long as a cultivator can go through the field and bury this fruit. Clean mulch, which keeps fruit off the ground, is also highly recommended. Removing leaves from the field as soon as the harvest season ends can significantly reduce the incidence of gray mold on fruit in June of the following year (Sutton et al., 1988). The use of tunnels—both high and low—can reduce the exposure to moisture and disease spores. The following biorational products are available commercially for Botrytis control: Serenade (Agraquest), Mycostop (Verdera Oy), and Promot (JH Biotech). Research in Israel found that combining two biocontrol agents (a yeast and bacterium) resulted in better suppression of Botrytis and reduced the variability of disease control (Guetsky et al., 2001). Although there is not a high level of gray mold resistance in any one strawberry cultivar, Earliglow is relatively resistant compared to most cultivars (Turns, 1990).
Leaf Spot
Leaf-spot diseases—identified by the presence of spots on strawberry leaves and stems—can be caused by the fungi Mycosphaerella fragariae, Ramularia tulasnei, or Phomopis obscurans, or by the bacterium Xanthomomas fragaiae. These pathogens are spread by splashing water and are harbored by dead leaves and other plant debris. Sanitation, as well as the recommendations mentioned on foliar disease preventatives, apply to leaf spot.
Please Note:
Preventive treatments such as sulfur, copper, or teas applied prior to wet weather are advisable for many diseases like leaf spot, gray mold, and powdery mildew. Also, studies have shown that systems using organic mulches have a reduced incidence of soil-borne pathogens.
Powdery Mildew
Powdery mildew is a fungal disease that affects strawberry foliage, flowers, and fruit. Caused by Sphaerotheca macularis, its spores prefer intermittently moist conditions and will not germinate in free-standing water. In coastal California strawberry fields, the practically year-round production season, foggy cool nights, and warm days make the disease a major and very persistent problem. Sulfur is the most common control agent on both conventional and organic farms. Milk has been used successfully against powdery mildew on cucurbit crops (Bettiol, 1999). Sonata™, a formulation of Bacillus pumilis, and Serenade (Bacillus subtilis) are approved for use on strawberries for powdery mildew control. Seven- to 14-day application intervals are recommended, depending on disease pressure. Other controls include horticultural oils, neem oil, and jojoba oil.
Greenhouse Production
Five factors—light, heat, pollination, pest control, and economics—make winter strawberry production in a greenhouse very different from field production.
Lighting is critical for winter production. The day-neutral cultivars (e.g., Tribute and Tristar) or the short-day types (e.g., Jewel) are much easier to grow during the short days of winter than most of the traditional June-bearing types. It is difficult and expensive to get the June-bearing types to fruit out of season. Even with the day-neutral types, some supplementary lighting will be necessary to get high-quality fruit.
Supplementary heat will have to be available (in some cases the lighting will provide enough heat). Although some non-fruiting vegetables (e.g., leafy greens, such as spinach) can produce well in unheated greenhouses, strawberry plants need about a 68º F day and 54º F night to produce good yields of high-quality berries.
The grower will have to provide pollination. Although self-fertilization in strawberry plants can occur, using bumble bees as pollinators for strawberries grown in greenhouses improves flower fertilization and, consequently, fruit quality, as well as the total fruit production (Dimou, et al., 2008). Various providers offer pollinator bees online.
Certain pests (usually the larger ones, e.g., tarnished plant bugs) can be effectively excluded from greenhouses, but others, such as mites, aphids, whitefly, thrips, and fungus gnats are likely to thrive and proliferate. Due to the need for bumble bees for pollination, controlling these pests with conventional pesticides is not a good idea. Fortunately, the pests can be effectively managed with biological controls, such as beneficial mites and lacewing larvae. For the details of greenhouse pest management, visit ATTRA’s Greenhouse Production topic page for our series of publications on greenhouse IPM.
Finally, prospective greenhouse growers should spend some time exploring local markets (restaurants, groceries, etc.). Off-season greenhouse growers will be competing with strawberries from California, Mexico, Chile, and Florida. Prices will have to offset the costs of production, so growers will have to produce an outstanding product. Check the Further Resources section for Greenhouse Strawberry Resources from the University of Massachusetts Amherst.
Tunnel Production

Photo: USDA/ARS
Tunnels can be low or high structures that provide season extension and crop protection from the elements. Unlike greenhouses, high tunnels are ventilated by raising the sidewalls or by opening the ends. Low tunnels are temporary structures usually one and a half to two feet tall, often constructed of wire hoops or PVC tubing. Low tunnels can be ventilated by simply uncovering the plants and placing the plastic off to the side while it is still fastened to the hoops.
Producers of day-neutral strawberries grown in tunnels have been able to extend production by warming and having the plants produce earlier and later in the season and by keeping rain off the fruit. These early and late periods of the season are when general supply is lower and prices are higher.
A high tunnel system is an increasingly popular conservation practice for farmers, and financial assistance is available through the Environmental Quality Incentives Program (EQIP).
Harvest and Postharvest

Photo: USDA/ARS
Strawberries must be picked and handled very carefully. The fruit must be firm, well-colored, and free from rot. When harvested at the right time and handled properly, strawberries will remain in good condition for many days. Most California- or Florida-grown strawberries found in supermarkets are picked three-quarters ripe to withstand shipping. The color of these strawberries is a full red, but the taste is disappointing. Small-scale growers who pick ripe fruit can easily compete with supermarket berries by offering a tastier, fresher berry to local consumers.
Proper postharvest handling of strawberries is essential. Cooling the berries will remove field heat and increase shelf life. Harvesting early in the day, while temperatures are cool, and then pre-cooling the fruit before shipping will extend the shelf-life significantly.
Forced-air cooling is the most common method used on strawberries. Flats are stacked parallel to each other in a cold room, with an open space between the flats. A tarp is then placed over the top and ends of the stacked cartons, with a fan located between stacks. The fan pulls cold air between the gaps of the stacked flats, removing the field heat from the berries. It is vital that the fruit be cooled as soon as possible. The more the delay between harvesting and cooling exceeds one hour, the greater the losses to deterioration (Kader, 1992). Water loss from strawberries can be a problem, so it is critical to maintain high humidity in the cooling facility. Avoid wetting the fruit, which can cause decay problems. Fresh-market strawberries are usually sold in one-piece molded-plastic containers called clamshells. The use of clamshells makes the strawberry pickers’ job a little easier; the wholesalers are not as concerned with the appearance of the fruit pack, since it looks uniform with the clear lid. Many of these clamshells are recyclable. A drawback to the clamshells is greater difficulty of cooling the fruit. The holes in the containers are not big enough to allow for rapid cooling, so extra time in the forced-air cooler is necessary.
Economics
Strawberries are one of the most popular fruits in the United States. They are a high-value crop, but they also have special production requirements, a short shelf-life, and a brief marketing season. Initial investment in land preparation, irrigation, and other equipment can be significant. Organic strawberries are in high demand, and this segment of the organic industry continues to grow at a rapid pace.
Continuous cropping of strawberries is not possible in an organic system that relies on crop rotations. The production cycle is shorter (one to two fruiting years) and yields may be both lower and more variable than in conventional systems. Labor requirements are another consideration in the cost analysis. Will there be enough pickers to keep up with peak production of the desired acreage?
Production-cost budgets for strawberries around the country are available through the following links:
-
- Sample Costs to Produce and Harvest Organic Strawberries. 2019. University of California Co-op Extension.
- Strawberry Economics: Comparing the Costs and Returns of Establishing and Producing Fresh and Processed Market June Bearing Strawberries in a Perennial Matted Row System to Day-Neutrals in a Perennial Hill, Plasticulture System, in the Willamette Valley. 2014. Oregon State University Extension Service. h
- Strawberry Growers Information: Budget/Cost Estimates. No date. North Carolina State Extension.
- Kentucky Matted Row Strawberry Profitability Estimated Costs and Returns. 2019. University of Kentucky.
- Strawberry Production: Sample Budget Worksheets. 2005. Penn State Extension.
- Florida Strawberry Production Costs and Trends. 2017. University of Florida IFAS Extension.
- Strawberry Production Costs in Greenhouse. 2015. University of Arizona CEAC Hydroponic Strawberry Information.
- 2011 Cost Estimates of Producing Strawberries in a High Tunnel in Western Washington. 2012. Washington State University Extension.
Marketing
Four basic marketing options are available to the strawberry grower: wholesale markets, cooperatives, processing firms, and direct sales to retail outlets or consumers. In wholesale marketing, either you or a shipper can take your crop to the market. Shippers generally sell and transport strawberries for a predetermined price. Wholesale marketing is subject to price fluctuations and is not usually very profitable, compared to direct marketing. Jim Cochran of Swanton Berry Farm in California, says, “I consider myself lucky to get five percent of gross. So, on a twenty-dollar flat of strawberries, (there is) a dollar for the company to keep.” (Inouye and Warner, 2001). Marketing cooperatives generally use a daily pooled cost and price, which spreads price fluctuations among all participating producers. Depending on your location and size, processors may or may not be a marketing option. Large-scale processors are less likely to contract with small-acreage growers; however, independent small-scale processing businesses often purchase from local growers.
Strawberries are successfully direct-marketed in a variety of ways, including farmers markets, roadside stands, and pick-your-own (PYO) operations. With pick-your-own operations, you save on harvest costs, but you must also be willing to accept some waste and higher liability-insurance costs. The trend in recent years has been toward an increase in prepacked strawberry sales at the farm and a reduced reliance on pick-your-own marketing (Fernandez and Hoffmann, 2012). When Pritts et al. compared the profitability of retailed berries picked by hired hands to marketing with a pick-your-own (PYO) strategy, profits were far lower in the PYO system (Pritts et al., 1999). Poor picking by inexperienced customers was assumed to reduce yield in the PYO by 10%. The PYO’s savings in harvest labor were not outweighed by the lower price charged to the consumer. Local restaurants and retailers such as grocery or health-food stores are another possible market, but you must take the time to contact produce managers and provide good-quality strawberries when stores require them. For more information on direct-marketing options, see the ATTRA publications Direct Marketing, Farmers Markets: Marketing and Business Guide, Community Supported Agriculture, Tips for Selling through CSAs, Selling to Restaurants, and Tips for Selling to Restaurants.
References
Achmon, Y., J.D. Fernandez-Bayo, K. Hernandez, D.G. McCurry, D.R. Harrold, J. Su, R.M. Dahlquist-Willard, J.J. Stapleton, J.S. VanderGheynst, C.W. Simmons. 2017. Weed seed inactivation in soil mesocosms via biosolarization with mature compost and tomato processing waste amendments. Pest Management Science. May p. 862-873
Anon. 1999. Science News. September 25. p. 207.
Arancon, N.Q., C.A. Edwards, P. Bierman, C. Welch, and J.D. Metzger. 2004. Influences of vermicomposts on field strawberries: 1. Effects on growth and yields. Bioresource Technology Vol. 93, No.2. p. 145-153.
Berry, R.E. 1998. Insects and mites of economic importance in the Pacific Northwest, 2nd edition. OSU Bookstore, Inc., Corvallis, OR. p. 74.
Bettiol, Wagner. 1999. Effectiveness of cow’s milk against zucchini squash powdery mildew (Sphaerotheca fuliginea) in greenhouse conditions. Crop Protection 18. p. 489-492.
Bolda M, L. Tourte, K Klonsky, et al. 2014. Sample Costs to Produce Organic Strawberries, Central Coast Region. UC Cooperative Extension.
Bomford, M.K. and R.S. Vernon. 2005. Root weevil (Coleoptera: Curculionidae) and ground beetle (Coleoptera: Carabidae) immigration into strawberry plots protected by fence or portable trench barriers. Environmental Entomology, Vol. 34, No. 4. p. 844-849.
Burrack, Hannah. 2017. It’s Time to Scout (And Manage) Spider Mites in Strawberries. North Carolina State Extension. Entomology- Insect Biology and Management.
Daar, S. 1988. Japanese beetles. Fine Gardening. May-June. p. 52–54.
Dara, S.K., and Sudha R. Dara. 2015. Soil application of the entomopathogenic fungus Metarhizium brunneum protects strawberry plants from spider mite damage. E-Journal of Entomology and Biologicals.
Demchak, K., T. Elkner, and M. Hu. 2019. Anthracnose on Strawberry Fruit. Penn State Extension Bulletin.
Dimou, M., Smaragda Taraza, Andreas Thrasyvoulou, and Miltiadis Vasilakakis. 2008. Effect of bumble bee pollination on greenhouse strawberry production, Journal of Apicultural Research. Vol. 47, No.2. p 99-101.
Dominguez, Pedro, Luis Miranda, Carmen Soria, Berta Santos, Manuel Chamorro, et al. 2014. Soil biosolarization for sustainable strawberry production. Agronomy for Sustainable Development. Vol. 34, No. 4. p.821-829.
Dufour, Rex. 2000. Farmscaping to enhance biological control. NCAT/ATTRA Pest Management Series. National Center for Appropriate Technology. p. 30.
Ellis, M. A., W. F. Wilcox, and L. V. Madden. 1998. Efficacy of metalaxyl, fosetyl-aluminum, and straw mulch for control of strawberry leather rot caused by Phytophthora cactorum. Plant Disease. Vol. 82, No. 3. p. 329-332.
Fernandez, G., and M. Hoffmann. 2012. Plasticulture Production – Plasticulture Information for N.C. Strawberry Growers. North Carolina State Extension.
Flint, M.L., and S.H. Dreistalt. 1998. Natural Enemies Handbook. The Illustrated Guide to Biological Control. Statewide Integrated Pest Management Project. University of California. Pub. 3386. p. 121.
Forcella, F., S.R. Poppe, N.C. Hansen, W.A. Head, E. Hoover and J. McKensie. 2003. Biological mulches for managing weeds in transplanted strawberry (Fragaria X ananassa). Weed Technology. Vol. 17, No. 4. p. 782-787.
Fuester, R.W., W.H. Day, C.H. Pickett, and K.A. Hoelmer. 2004. Introduction, release, and establishment of European Peristenus Spp. on mirid plant pests in North America. Proceedings of the 15th International Plant Protection Congress, Beijing, China, May 11-16. p. 132.
Gamliel, A., and J.J. Stapleton. 1997. Improvement of soil solarization with volatile compounds generated from organic Amendments. Phytoparasitica. Vol. 25, S31-S38.
Gaskell, M. 2004. Nitrogen availability, supply, and sources in organic row crops. p. 13-20. California Conference on Biological Control CCBC IV. In: Proceedings of California Organic Production and Farming in the New Millennium: A Research Symposium. International House, Berkeley, California.
Guetsky, R., D. Shtienberg, Y. Elad, and A. Dinoor. 2001. Combining biocontrol agents to reduce the variability of biological control. Phytopathology. Vol. 91, No. 7. p. 621-627.
Guthman, Julie. 2017. Land access and costs may drive strawberry growers’ increased use of fumigation. California Agriculture. Vol. 71, No. 3. p. 184-191.
Hagler, J.R., D.J., Nieto, S.A. Machtley, D.W. Spurgeon, B.N. Hogg, and S.L. Swezey. 2018. Dynamics of Predation on Lygus hesperus (Hemiptera: Miridae) in Alfalfa Trap-Cropped Organic Strawberry. Journal of Insect Science (Online). Vol. 18, No. 4, p. 12.
Handley, D.T., A. Wheeler, and J.F. Dill. 2002. A survey of strawberry inflorescence injury caused by the strawberry bud weevil. Strawberry research to 2001. Proceedings of the 5th North American Strawberry Conference, 2002. p. 82-84.
Handley, D.T., J.F. Dill, and J.E. Pollard. 1991. Field susceptibility of twenty strawberry cultivars to tarnished plant bug injury. Fruit Varieties Journal. Vol. 45, No. 3. p. 166.
Hartz, T.K., J.E. DeVay and C.I. Elmore. 1993. Solarization is an effective solar disinfestation technique for strawberry production. HortScience. Vol. 28. p. 104-106.
Huang, Xinqi, Teng Wen, Jinbo Zhang, Lei Meng, Tongbin Zhu, and Zucong Cai. 2015. Toxic organic acids produced in biological soil disinfestation mainly caused the suppression of Fusarium oxysporum f. sp. Cubense. BioControl. February. p. 113-124.
Inouye, Janel, and Keith Douglass Warner. 2001. Plowing Ahead: Working Social Concerns into the Sustainable Agriculture Movement. California Sustainable Agriculture Working Group White Paper. Santa Cruz, California.
Johnson, M.S., and S.A. Fenimore. 2005. Weed and crop response to colored plastic mulches in strawberry production. HortScience. Vol. 40, No. 5. p. 1371–1375.
Kader, A.A. 1992. Postharvest technology of horticultural crops. Second edition. Publication 3311. University of California. p 227.
Kovach, J., W. Wilcox, A. Agnello, and M. Pritts. 1993. Strawberry IPM Scouting Procedures. New York State Integrated Pest Management Program. Publication 203b. Cooperative Extension Service, Cornell University, Ithaca, NY.
Kovach, Joe, and Greg English-Loeb. 1997. Testing the efficacy of Mycotrol ES, Beauveria bassiana, on tarnished plant bugs, Lygus lineolaris, in New York strawberries.
Maas, J.L. (ed.). 1987. Compendium of Strawberry Diseases. American Phytopathological Society, St. Paul, Minnesota.
Momma, Noriaki. 2008. Biological soil disinfestation (BSD) of soilborne pathogens and its possible mechanisms. Japan Agricultural Research Quarterly. Vol. 42, Issue 1. p. 7-12.
Pinkerton, J.N., K.L. Ivors, P.W. Reeser, P.R. Bristow, and G.E Windom. 2002. The use of soil solarization for the management of soilborne plant pathogens in strawberry and red raspberry production. Plant Disease. Vol. 86, No. 6. p. 645-651.
Pritts, M.P., M.J. Kelly and G. English-Loeb. 1999. Strawberry cultivars compensate for simulated bud weevil damage in matted row plantings. HortScience. Vol. 34, No. 1. p. 109-111.
Pritts, M. 2002. A future for the perennial matted row? The Berry Basket. Vol. 5, No. 1 p. 11.
Strand, Larry L. 1993. Integrated Pest Management for Strawberries. Publication 3351. University of California. p. 15.
Subbarao, K.V., J.C. Hubbard, and S.T. Koike. 1999. Evaluation of broccoli residue incorporation into field soil for Verticillium wilt control in cauliflower. Plant Disease. Vol. 83. p. 124-129.
Sutton, J.C., T.D.W. James, and A. Dale. 1988. Harvesting and bedding practices in relation to grey mould of strawberries. Annals of Applied Biology. Vol. 113. p. 167-175.
Swezey, S. 2004. Trap cropping the western tarnished plant bug, Lygus Hesperus Knight, in California organic strawberries. In: Proceedings, California Organic Production and Farming in the Millennium: A Research Symposium. July 15, 2004. International House, Berkeley, California.
Tilmon, K.J. and M.P. Hoffmann. 2003. Biological control of Lygus lineolaris by Peristenus spp. in strawberry. Biological Control. Vol. 26, No. 3. p. 287-292.
Testen, L. Anna and Sally A. Miller. 2017. Anaerobic Soil Disinfestation for Management of Soilborne Diseases in Midwest Vegetable Production. Ohio State University Extension, HYG-3315.
Turns, E.E. 1990. Strawberry breeding has many “ifs.” American Fruit Grower. February 1990. p. 48.
Udayagiri, S.S.C. Welter, and A.P. Norton. 2000. Biological control of Lygus hesperus with inundative releases of Anaphes iole in a high cash value crop. Southwestern Entomologist Supplement 23. p. 27.
van Agtmaal, Maaike, Gera J. van Os, W.H. Gera Hol, Maria P.J. Hundscheid, Willemien T. Runia, Cornelis A. Hordijk, and Wietse de Boer. 2015. Legacy effects of anaerobic soil disinfestation on soil bacterial community composition and production of pathogen-suppressing volatiles. Frontiers in Microbiology. July.
Weber, C.A. 2003. Biodegradable mulch films for weed suppression in the establishment year of matted-row strawberries. Hortechnology. Vol. 13, No. 4.p. 665–668.
Welke, S.E. 2004. The effect of compost extract on yields of strawberries and the severity of Botrytis cenerea. Journal of Sustainable Agriculture. Vol. 25. No.1. p. 57-68.
Further Resources
2016 Organic Production and IPM Guide for Organic Strawberries. Cornell University Cooperative Extension. NYS IPM Publication No. 226.
Current and Future Prospects for Biodegradable Plastic Mulch in Certified Organic Production Systems. 2019. eOrganic.
Greenhouse Strawberry Resources. University of Massachusetts Amherst. The Center for Agriculture, Food and the Environment.
Organic Strawberry Growing: The Ultimate How-To Guide. YouTube video. Urban Farmer Curtis Stone.
Sustainable Practices for Plasticulture Strawberry Production in the Southeast. 2020. North Carolina State Extension Publication.
Vendors of Beneficial Organisms in North America. 2010. By Jen White and Doug Johnson. University of Kentucky ENTFACT-125.
Strawberries: Organic Production
By Martin Guerena, NCAT Agriculture Specialist
Published 2007, updated April 2021
© NCAT
IP046
Slot 39
Version 042821
This publication is produced by the National Center for Appropriate Technology through the ATTRA Sustainable Agriculture program, under a cooperative agreement with USDA Rural Development. ATTRA.NCAT.ORG.
Related Publications
- A Pictorial Guide to Hedgerow Plants for Beneficial Insects
- Biointensive Integrated Pest Management
- Companion Planting & Botanical Pesticides: Concepts & Resources
- Farmscaping to Enhance Biological Control
- Tree Fruits: Organic Production Overview
- Planning for Profit in Sustainable Farming
- Sustainable Management of Soil-Borne Plant Diseases
- Soil Solarization and Biosolarization


