Parasite Management for Natural and Organic Poultry: Coccidiosis
By Anne Fanatico, NCAT Agriculture Specialist
Abstract
As the size of outdoor flocks increases, more attention is needed for coccidiosis control. Coccidia are parasites that damage the gut of poultry. Photo: Joe Beasley, DVM, PhD
Both small and large poultry producers are interested in the sustainable management of the parasitic disease coccidiosis. This publication provides information on its life cycle, transmission in free-range production, management in the brooder and on pasture, natural treatments, drugs, and vaccines. On a small scale, coccidiosis can be handled without medication by careful management, especially during brooding, and adequate pasture rotation; however, on a larger scale, it is more difficult and vaccines are an important alternative to drugs in organic production. References and further information follow the narrative.
Contents
- Introduction
- Life Cycle and Types of Coccidia
- Transmission in the Environment
- Symptoms and Diagnosis
- Management for Control
- Natural Treatments
- Drugs
- Vaccines
- Summary
- References
- Further Resources
Introduction
In the past, coccidiosis was one of the diseases most feared by commercial poultry growers in the U.S. Death losses of 20 percent or more were common. ‘Backyard’ growers are usually so small that coccidiosis is not a problem, but as the size of free-range flocks increases, coccidiosis becomes a threat.
Small producers in the U.S. raise birds with outdoor access and sell the meat and eggs directly to local consumers. These ‘pastured poultry’ flocks are increasing in number and size. Many of these producers use natural production methods and avoid using drugs in their flocks. Larger companies also produce certified organic poultry under the USDA National Organic Program rules, which do not permit the use of anticoccidial drugs.
The conventional poultry industry is likewise interested in reducing its reliance on drugs. The industry raises poultry on a large scale with high-density flocks. Coccidiosis is controlled with preventative drugs. In fact, high-density production became possible only after the development of preventative anticoccidial drugs in the 1940s. However, coccidia are becoming increasingly resistant to drugs, and the poultry industry is looking for alternatives. The use of vaccines in particular holds potential for both small and large growers.
Coccidiosis is a parasitic disease that can cause severe losses in poultry meat and egg production. The parasites multiply in the intestines and cause tissue damage, lowered feed intake, poor absorption of nutrients from the feed, dehydration, and blood loss. Birds are also more likely to get sick from secondary bacterial infections. However, in low-density production or with the use of preventative medication, coccidiosis generally remains a subclinical disease that only affects performance – without the alarming losses of the past.

Posture of sick birds. Photo: Lloyd Keck, DVM
Producers used to dread outbreaks of bloody diarrhea. An outbreak of coccidiosis left untreated, eventually runs its course, and most of the flock will survive. The birds that recover from coccidiosis gain immunity, but production may never recover. If the infection is severe, the gut remains scarred and impaired, and stunted broilers do not catch up in weight gain.
The production system and the stocking density have a significant impact on coccidiosis. Low-density production systems allow a low level of exposure in which immunity develops without making the birds sick and damaging performance. Birds are then protected. However, as the size of flocks increase, the numbers of coccidia also grow and can pose a threat to the flock.
Life Cycle and Types of Coccidia
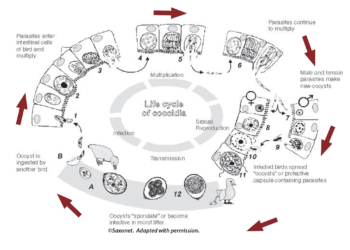
Knowing how coccidia develop helps to understand and control the disease. Coccidiosis is caused in poultry by a one-celled parasite of the genus Eimeria. The life cycle of Eimeria takes about four to seven days to complete. It begins when active ‘oocysts’ are picked up by the bird and swallowed. An ‘oocyst’ is a capsule with a thick wall protecting the parasites. They ‘sporulate’ or become infective if moisture, temperature, and oxygen become conducive to growth. After a bird eats the oocysts, coccidia imbed in the intestinal lining and multiply several times, damaging tissue.
A coccidial infection differs from bacterial and viral infections because coccidia are ‘self-limiting’ and usually stop multiplying before killing the bird.
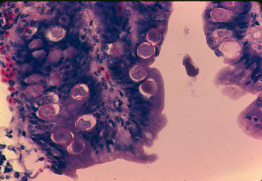
Coccidia multiply in intestinal cells. Photo: Joe Beasley, DVM, PhD.
Coccidia are parasites, so they get their nutrients from the chicken host. The multiplications eventually stop, usually before causing death of the bird. The bird sheds the parasite in its droppings. These new oocysts can infect other birds. See the box ‘Coccidial Multiplication’ for further details on coccidia’s complex life cycle.
Coccidiosis is usually a disease of young birds, but birds can be infected at any time if never before exposed. Coccidia populations take time to build to dangerous levels, therefore outbreaks usually occur when birds are between 3 and 8 weeks of age. Coccidiosis goes hand-in-hand with gut diseases, because it damages the gut and allows bacteria to enter and cause secondary infections. Coccidia are ‘species-specific’ – coccidia that affect chickens do not affect other livestock, and vice versa (see Species-Specific Parasites box).
Coccidial Multiplication
Coccidia are very prolific parasites. A single sporulated oocyst can have a big impact when eaten by a chicken. Each oocyst has four sporocysts in it, and each sporocyst has two sporozoites in it. The digestive tract releases the eight sporozoites from the oocyst, and they move into the cell lining of the digestive tract. Inside the cell, the parasite divides and invades more cells. There may be several generations of asexual multiplication; however, this stage is self-limiting and eventually stops. Finally, a sexual stage occurs in which male and female organisms unite and form new oocysts that are protected by a thick wall. These oocysts are shed in the feces. See Coccidia Life Cycle Diagram. For here more detailed information.
Species-Specific Parasites
Almost all livestock are affected by different types of coccidia. Each type of coccidia infects only one species of livestock—each is “speciesspecific.” There are seven different Eimeria that infect chickens, but only three cause most of the trouble in the U.S.: Eimeria tenella, Eimeria maxima, and Eimeria acervulina. Immunity to one type does not provide immunity for other types. Turkeys, ducks, geese, and other types of poultry are all infected by different types of coccidia.
Chicken coccidia species:
Eimeria acervulina
Eimeria maxima
Eimeria tenella
Eimeria necatrix
Eimeria mitis
Eimeria brunetti
Eimeria praecox
Turkey coccidia species:
Eimeria adenoeides
Eimeria meleagrimitis
Eimeria gallopavonis
Eimeria dispersa
Transmission in the Environment
Chickens get coccidiosis by eating oocysts that have been shed in the droppings of infected chickens. Infected chickens shed oocysts for several days or weeks. Oocysts sporulate within two days under the proper conditions and become infective. Chickens pick them up by pecking on the ground or in litter used for bedding in the house. Oocysts can also be spread by insects, dust, wild birds, and humans (from shoes and equipment).
Oocysts can survive many weeks in the soil outdoors – as long as 600 days. (Farr and Wehr, 1949) The optimum temperature for sporulation is around 72°F. The rate of sporulation is slower if temperatures are much cooler or hotter. Oocysts are killed either by freezing or very high temperatures.

Chicks can pick up oocysts both indoors and on pasture. Photo: NCAT
Sporulation also requires oxygen and moisture (at least 20 percent moisture in the litter for optimal sporulation). If the litter feels damp to the back of your hand, it is damp enough for sporulation. Once sporulated, the oocyst remains infective for months if protected from very hot, dry, or freezing conditions.
In very large poultry houses, oocysts do not last long in the litter because of the action of ammonia released by decomposition of litter and manure and by the action of molds and bacteria. However, there are usually so many oocysts that birds continue to pick them up and get sick.
Symptoms and Diagnosis
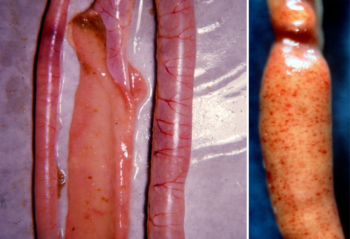
Small intestines affected by Eimeria maxima. The middle sample is opened to show white spots (left photo). Photo on right shows ballooning. Photos: Lloyd Keck, DVM
Outward signs of coccidiosis in chickens include droopiness and listlessness, loss of appetite, loss of yellow color in shanks, pale combs and wattles, ruffled, unthrifty feathers, huddling or acting chilled, blood or mucus in the feces, diarrhea, dehydration, and even death. Other signs include poor feed digestion, poor weight gain, and poor feed efficiency. Some symptoms can be confused with other diseases. For example, necrotic enteritis is a gut disease that also causes bloody diarrhea.
Producers in the past identified coccidiosis outbreaks as either severe-acute or chronic, which was less severe but more widespread.
If concerned about coccidiosis, do a necropsy – put on plastic gloves and cut open the chicken. Look at the intestines and then cut them open. If done soon after death, it may be possible to identify characteristic lesions or sores in the gut. Coccidiosis causes a thickening of the intestines, which make them feel like a sausage. There may be light-colored spots on the surface of the gut, and inside the gut, hemorrhages and streaks. If you want to confirm a diagnosis, you can send scrapings of the gut lining to a state diagnostic lab. The USDA’s Animal and Plant Health Inspection Service’s Website lists diagnostic labs.
The type and location of lesions in the gut indicates the species of Eimeria.
- Eimeria acervulina affects the upper part of the small intestine. You may see small red spots and white bands on it.
- Eimeria maxima affects the entire small intestine. The intestines look watery, and in later stages have blood and mucus. The intestine may look thickened and ballooned with red pinpoint lesions.
Eimeria tenella affects the blind sacs (ceca) of the gut. They may be filled with blood and pus and turn into a solid core.
Management for Control
Management has always been important to coccidiosis control, especially before drugs were available. Management focuses on reducing the number of coccidia to keep infection at a minimum until immunity is established.
Natural Immunity
A small-scale, low-density production system can allow a low level of exposure to coccidia, which permits the chick to develop immunity without triggering the disease. However, birds may not pick up enough parasites to cause immunity, or they may be overwhelmed by too many. In addition, immunity is only species-specific. Exposure to one type of coccidia will not protect a chicken from the other six types that can infect it.
Early detection is a management method to avoid the use of preventative medication. If you can catch the disease when it initially infects only a few birds, you may have time to treat the birds with a rescue drug or make a management change, such as moving the birds to fresh pasture. Early detection requires close observation and experience. Watch feed intake in particular – it goes down in the early stages of coccidiosis.
The choice of production system is an important management decision. High-density, large-scale production almost always requires the use of anticoccidial medication. In contrast, in low-density, small-scale production, the birds tend to stay ahead of the parasites and may not require medication. Many small-scale producers do not use anticoccidial medication; however, as the size of the flocks grows, more problems are encountered and more management is required for natural immunity.
Immunity is especially important in turkeys, layers, breeders, and slow-growing broilers that are kept longer than fast-growing broilers marketed at a younger age.
Small-Scale Poultry Production Systems
Small poultry producers often provide outdoor access with either a permanent house and yard or portable houses. Small pens that are moved daily are also used.
These small growers usually brood chicks in a separate area before moving them to the outdoor facility for growout. However, some growers brood chickens in the same house in which they are grown.

House with yard. Photo: NCAT

Small pen moved to fresh pasture. Photo: NCAT

Portable houses with fence. Photo: NCAT
Brooder and Growout Management
When chicks are brooded in a separate area before moving them to the growout facility (two-stage production), the brooder stays clean of infective oocysts since fast-growing broilers do not remain past three weeks of age. However, chicks are at risk for coccidiosis if they stay in the brooder longer than three weeks. Pullet chicks for egg laying grow slowly and stay in the brooder longer. If chicks are brooded and grown out in the same facility (one-stage production), they seed the area with coccidia. These birds may require a lower density or, possibly, medication. The following management strategies for good brooding can help.
Good brooding practices can reduce the need for medication and include not only sufficient space but also sanitation and litter management. Give birds adequate floor space and feeder/waterer space to prevent overcrowding. Small flock producer Robert Plamondon recommends at least one square foot of floor space per chick and four tube feeders per 100 chicks. (Plamondon, 2003)

The chicks are kept in this brooder for only a few weeks and later moved to a growout pen or small house. Photo: NCAT
Keep the feeders full. If feeders go empty, birds forage in the litter and ingest oocysts. The longer they peck at contaminated litter, the more oocysts they will ingest.
Sanitation
Disinfectants are not effective against coccidia, so sanitation focuses on good hygiene and removing infected droppings.
- Put waterers and feeders at a height level with the backs of the birds, so they cannot defecate or scratch litter into them. Keep birds from roosting on the feeders with anti-roosting wire. Suspend waterers or put them on wire-covered platforms to help keep them clean.
- Clean the waterers and feeders frequently.
- Keep older birds away from chicks, since old birds are carriers.
- Add fresh litter or rake litter frequently to cover parasites.
Litter Management
Keep the litter dry to reduce sporulation of oocysts. Remove any wet or crusted litter. Moisture in the litter is affected by the following:

Keep litter dry by preventing water spills. Photo: NCAT
- Heat source: A propane radiant brooder heats a larger area and dries out litter more than a heat lamp.
- Ventilation: Housing should prevent drafts but not be airtight. Humidity, along with ammonia and other gases, needs to escape.
- Water leaks: Water leaks must be prevented.
- Condensation: Condensation may occur in buildings with uninsulated roofs and walls and will contribute to litter moisture.
- Feed: Rations with excessive protein or excessive salt can result in wet litter.
In the large-scale industry, ‘new-house coccidiosis syndrome’ sometimes occurs when birds are placed on brand-new litter. There is no low-level population of coccidia to establish immunity, so the flock is more susceptible, coccidiosis problems are more likely, and medication may be needed.
Some small flock producers are interested in the built-up or composting litter as an ecosystem of microbes.
Poultry-house litter becomes significantly anti-coccidial after about six months’ use, as organisms that eat coccidia start to thrive and knock down the coccidia population… By never removing more than half the brooder house litter at a time, it can keep its anti-microbial properties indefinitely. (Plamondon, 2002a)
Plamondon recommends starting with at least six inches of shavings and adding a thin layer of fresh litter on top, which will prevent chicks from eating old litter at first. He turns it daily with a spading fork to keep it from getting packed down and crusted over. If the litter seems too wet, he adds more dry litter. He only removes litter when it is too deep to manage or when too wet.
(Plamondon, 2002b)
Also, although oocysts can be destroyed by microbes in the litter and soil, there may be so many oocysts that the birds become infected. Unfortunately, there is little scientific information available on composting litter.
Pasture
Producers provide outdoor access to allow poultry to express natural behavior, increase space, and to provide fresh air and sunlight. Outside, birds may pick up fewer oocysts, since they are more likely to peck forage instead of droppings; however, access to the outdoors has both advantages and disadvantages for coccidial control. Extreme heat and cold outdoors can reduce sporulation or kill oocysts. Yet warmth and moisture are favorable conditions for coccidia. Before the use of medication, commercial producers used to experience coccidiosis outbreaks in late spring, summer, and early fall.
In the warm, humid South, coccidiosis is a greater problem than in dry western states. Dry conditions on pasture greatly reduce coccidiosis. In cold areas, although oocysts on pasture may die during winter, the chickens in the house during winter still carry oocysts and reseed the pasture with them in the spring.
It is important to control areas of high traffic outdoors to reduce the number of oocysts. The locations of the waterers and feeders, the pasture, and the house itself, if possible, should be rotated. Straw, litter, or bark can help control muddy areas. Controlling coccidiosis on pasture is trickier with broilers than with layers, since the broilers are faster-growing and less active. They eat a lot, generating large amounts of manure, and congregate in shaded areas. Layers get off the ground to roost on perches. Keeping birds in a floorless pen that is moved daily eliminates coccidiosis by breaking the life cycle – oocysts cannot re-infect birds.
Natural Treatments
Keeping birds in general good health is always important. Some small producers provide raw milk, yogurt, apple cider vinegar, or probiotics to birds, believing that beneficial microbes will prevent or treat coccidiosis. Actually, coccidia do not compete with bacteria in the gut; therefore, beneficial bacteria and other microbes will not eliminate coccidial development. However, anything that improves the overall health of the gut and the bird can help reduce the impact of coccidiosis. Also, a population of beneficial bacteria is always better than pathogenic bacteria, since coccidia weaken the gut wall, and bacteria may pass through. In short, feeding dairy products or probiotics will not stop the coccidia through ‘competitive exclusion’ but does provide nutrients or beneficial bacteria that are useful in any situation.
Producers sometimes give diatomaceous earth (DE) to the birds in the belief that the sharp edges of the fossilized diatoms will damage the parasites and reduce coccidiosis; however, there is no scientific data to support its use.
Drugs
Drugs are used for two different purposes:
- To prevent illness
- To treat illness
Although a producer may depend on management for coccidiosis control, a drug such as amprolium is useful for rescue treatment in the case of an outbreak. There is no need to destroy infected birds; they can be treated. In large houses, it is necessary to routinely use drugs or vaccines because of the high density of birds.
Types of Drugs
- Sulfa drugs: An exciting discovery in the 1930s was that sulfa drugs would prevent coccidiosis – the first drugs shown to do so. Sulfa drugs also have some antibacterial action. However, a relatively large amount of sulfa was needed (10-20 percent of the diet) and could be tolerated by the bird for only a short time, since it caused rickets. (Reid, 1990) Sulfa drugs had to be used intermittently (e.g., three days on and three days off). Nowadays, comparatively small amounts of sulfamonaides, such as sulfaquinoxaline, are used. They work only against Eimeria acervulinaand Eimeria maxima, not against Eimeria tenella. Sulfamonaides are used to treat coccidiosis.
- Amprolium: Amprolium is an anticoccidial drug. It has also been used for many years and needs no withdrawal time to guard against residue in the meat. It is given in the drinking water and interferes with metabolism of the vitamin thiamin (vitamin B1) in coccidia. Amprolium treats both intestinal and cecal coccidia.
- Quinolones: Quinolones are ‘coccidiostats’ that arrest the coccidia in an early stage of development. An example is decoquinate (Deccox®). The drugs are used for prevention.
- Ionophores: Ionophores are anticoccidials commonly used in the large-scale industry. They alter the function of the cell membrane and rupture the parasite. Ionophores also have antibacterial action and help prevent secondary gut diseases. Ionophores are not synthetic drugs; they are produced by fermentation and include monensin (Coban®) and salinomycin (Sacox®). However, some ionophores are now completely ineffective against coccidia because of resistance the coccidia have developed. They are used for prevention.
- Other drugs: There are many other anticoccidial drugs in various chemical classes with various modes of action. Examples are Nicarb® (nicarbizone) and Clinicox®.
Using Drugs
You need a veterinarian’s prescription to use drugs for poultry (but not to use vaccines). Feed mills need a license to put drugs in feed.
In the large-scale industry, drugs are used for prevention rather than treatment. If you treat the bird after an outbreak, the damage is already done. Many preventative drugs are effective only in the first part of the parasite life cycle, and therefore must be used early if they are to be used at all.
Drawbacks of using preventative drugs are their expense and he resistance that coccidia have developed. The drugs are not as effective now as when they were first introduced. Large companies use a drug rotation or shuttle program to reduce resistance.
There are not many new anticoccidial drugs because of the extensive process for FDA approval. It costs millions of dollars to develop a new anticoccial drug and get it approved for use.
In the large-scale industry, most anticoccidial drugs are withdrawn a week before slaughter of broilers to save money or to prevent residue in the meat. Drugs are withdrawn before layers begin laying eggs to prevent residues in the eggs.
Small producers often give pullets medicated feed while in the brooder, and then remove medication when they are older and placed in pasture-based systems.
Unfortunately, drugs used for prevention usually interfere with development of immunity to coccidia. On the other hand, using drugs for treatment only does allow immunity to develop. If signs of the disease appear, use drugs that are appropriate for coccidia’s late life cycle – only sulfonamides and amprolium. (Reid, 1990) When birds are getting sick, they lose their appetite. Therefore, soluble medication should be provided in the drinking water.
Vaccines
Interest is growing in controlling coccidiosis by vaccination because immunological control is recognized as the only practical alternative to anticoccidial drugs in largescale production. (Chapman, 2002)
Large poultry companies usually vaccinate chicks at company-owned hatcheries. Smaller producers buy chicks from independent hatcheries, but some hatcheries do not offer coccidiosis vaccination. Small producers may need to do the vaccination themselves, once the chicks arrive at the farm.
Types of Vaccines
At the time of this writing (2006), coccidial vaccines licensed in the U.S. include the following:
- Coccivac®: This vaccine was developed in the early 1950s. The ‘B’ and ‘D’ types are different mixtures of Eimeriaspecies; the ‘T’ type is for turkeys. Coccivac® is produced by Schering Plough Animal Health.
- Immucox®: This vaccine was developed in Canada by Vetech Laboratories. It is distributed by Wingo.
- Advent®: This vaccine was recently developed in the U.S. by Viridus Animal Health. It is marketed as having more viable oocysts (truly sporulated oocysts that can cause immunity) than other vaccines.
The vaccines above can actually cause some lesions and occurrence of coccidiosis in birds because they are not ‘attenuated’ or weakened in some way. It is a controlled occurrence, but it may be necessary to treat for secondary gut disease, using antibiotics or alternatives such as probiotics. In contrast, coccidiosis vaccines used in Europe are attenuated. They are altered because the coccidia used in the vaccine are designed to mature quickly and have a short (‘precocious’) life cycle and low fertility. They are not pathogenic – disease-causing – and are more costly to produce than the nonattenuated vaccines. They include Paracox®, Livacox®, and Viracox® which are marketed in other countries but not currently in the U.S.
More types of vaccines are likely to be developed, because the government approval process is much cheaper for vaccines than for anticoccidial drugs.
Since immunity is species-specific, anticoccidial vaccines include mixtures of species of Eimeria that affect chickens. It is especially important to include the three types that cause the most damage in chickens: Eimeria acervulina, Eimeria maxima, and Eimeria tenella.
Using Vaccines
Birds need good protection by the time they are three weeks old, so vaccines should be given at the hatchery or by one week.
Methods of application:
- Spray cabinets: These are used at hatcheries on day-old chicks and may include a dye to indicate application. This is the most uniform method of application, resulting in 90 to 95 percent of chicks exposed to the vaccine. (Chapman, 2000)
- Edible gel: Gel pucks are placed in transport crates or on the floor of the house when the chicks arrive. The gel is brightly colored to attract the attention of the chicks. Immucox ® is administered in this form.
- Feed spray: Vaccines are mixed with water in a garden pressure-sprayer and sprayed on a 24-hour supply of feed. Advent® is either sprayed on the feed in this manner or applied in a spray cabinet at the hatchery.
- Drinking water: The chicks should be slightly water-starved to encourage them to drink. Since oocysts are heavy and fall to the bottoms of drinkers, they are mixed with a suspension agent to keep them evenly distributed. (Chapman, 2000) This method can be used for older chicks. Vaccines cannot be given through proportioners or nipple drinkers.
To confirm the method of application, check the tag. For example, Coccivac® is given to turkey poults by spray cabinet at 1 day old; feed spray at 1 to 3 days old, and via drinking water from 3 to 14 days old.
It is important to apply vaccines uniformly to ensure the birds get equal exposure. If birds receive too much of a nonattenuated vaccine, the parasites can cause lesions. If attenuated vaccines are not given in adequate doses, the birds will be susceptible to field strains of the coccidia.
(Chapman, 2000)
The environment must allow the oocysts to sporulate, since the goal of vaccination is to introduce the parasite in small numbers. Litter should be damp but not wet. (Chapman, 2000) After vaccination, birds excrete fresh oocysts onto the litter. Birds then eat these (second cycle) oocysts. (Chapman, 2000) Two cycles of replication are needed for good protection.
Vaccines are usually sold only in large amounts. Advent is sold in 1,000-dose vials that cost about $13.50 each and must be purchased in boxes of 10 vials per box. Immucox can be purchased in smaller amounts. A tube of gel costs about $70 and has 28 ‘slices.’ Each slice serves 100 birds. Half tubes can also be purchased for about $35.
Since the vaccines contain live oocysts, they should not be frozen. Birds need access to their droppings in order for the vaccine to work, since oocysts must be reingested. Vaccines are not effective for birds raised in batteries or cages with wire floors.
Vaccines have been used for some time to provide immunity for broiler breeders and commercial egg layers, but there is less use in broilers.
Vaccine boosters are not normally given. Broilers usually have a short life and do not need boosters. Longer-lived birds like layers are constantly re-exposed to coccidia, so immunity is topped off constantly.
Do not give drugs and vaccines to the same flock – they are opposed to each other.
If your flock is raised under intensive conditions, you will eventually need to vaccinate or use drugs. Tips for using vaccines in large-scale production are provided below. Although most large poultry companies work with their veterinarians on health issues, these tips will be useful.
Tips for Using Vaccines in Large-scale Production
‘Partial house’ brooding and brooder rings can cause problems for coccidiosis management in large-scale production. In partial-house brooding, part of the house is sectioned off for brooding, while the rest of the house is vacant. Therefore, the whole house is not seeded with a low level of oocysts to help establish immunity. Producers should either release broilers to the whole house before 6 days of age or hold them until 12 days. (Chapman, 2002)
‘Chuck Martin of Vetech Laboratories Inc. find their customers achieve the best results if when brooding they provide .5 square foot per bird to day seven, then increase by .5 square foot per bird each week for the next couple weeks.’
Although organic companies cannot use routine drugs, it is important to know about drug/vaccine interactions. The conventional poultry industry uses vaccines in combination with drugs to help deal with the problem of drug resistance in broilers. Sensitivity testing can determine the level of resistance. Vaccines are prepared with coccidia strains that are susceptible to drugs. The vaccinal coccidia reproduce with field-strain coccidia in the broiler house and transfer drug susceptibility to drug-resistant strains. Vaccines essentially seed a house with coccidia that are sensitive to drug use. Therefore, large producers rotate drugs with vaccines to restore drug sensitivity to the field strains or replace the field strains with drug-sensitive strains.
Since the nonattenuated (unweakened) vaccines can cause lesions, pathogenic bacteria can enter the gut. You may need to treat for necrotic enteritis at 16 to 17 days with an antibiotic. (Chapman, 2002) These secondary bacterial infections are a particular problem for organic companies. Treating birds with antibiotics for necrotic enteritis means removing them from the organic program. Fortunately, there are natural alternatives to antibiotics, such as probiotics. It is also important to control factors that predispose birds to necrotic enteritis, such as feeding fishmeal. Poor uniformity of vaccine application and partial-house brooding or brooder rings can increase the incidence of complications.
Summary
Small-scale producers can control coccidiosis with a good understanding of its life cycle and conditions for transmission, by management strategies such as good litter and pasture rotation, and by using drugs only for rescue, if needed. Large-scale producers, especially organic producers, increasingly rely on vaccines.
References
Chapman, D. 2000. Practical use of vaccines for the control of coccidiosis in the chicken.
World’s Poultry Science Journal. Vol. 56. p. 7-12.
Chapman, D. 2002. Sustainable coccidiosis control in poultry production: The role of live vaccines. International Journal for Parasitology. Vol. 32. p. 617- 629.
Farr, M.M., and E.E. Wehr. 1949. Survival of Eimeria acervulina, E. tenella, and E. maxima oocysts on soil under various field conditions. Annual N.Y. Academy of Science. Vol. 52. p. 468-472.
Plamondon, Robert. 2003. Re: Probiotics (Also Coccidiosis). E-mail posting to PasturePoultry listserver. March 1.
Plamondon, Robert. 2002a. Re: Coccidiosis. E-mail posting to PasturePoultry listserver.
December 20.
Plamondon, Robert. 2002b. Coccidiosis control. Email posting to PasturePoultry listserver. April 3.
Reid, Malcolm, W. 1990. History of avian medicine in the United States. X. Control of coccidiosis. Avian Diseases. Vol. 34. p. 509-525.
Further Resources
Buvanderan, V., and P. Kulasegaram. 1972. Resistance of breeds and breed crosses of chickens to experimental Eimeria necatrix infection. British Veterinary Journal. Vol. 128. p. 177-183.
Long, P.L. 1968. The effect of breed of chickens on resistance to Eimeria infections.
British Journal of Poultry Science. Vol. 9. p. 71-78.
Merck Manual On-Line
www.merckvetmanual.com/mvm/index.jsp
Organic Livestock Research Group. 2000. Coccidiosis. In: Poultry Health and Welfare in Organic Farming. Veterinary Epidemiology and Economics Research Unit ( VEERU), Department of Agriculture, The University of Reading, U.K.
Parasite Management for Natural and Organic Poultry: Coccidiosis
By Anne Fanatico
NCAT Agriculture Specialist
IP245
This publication is produced by the National Center for Appropriate Technology through the ATTRA Sustainable Agriculture program, under a cooperative agreement with USDA Rural Development. ATTRA.NCAT.ORG.